The Scaling Gap: What’s Holding Back Digital Behavioral Health
April 20, 2026
Background
VEXAS syndrome is a rare disorder that affects adults and is caused by mutations in the UBA1 gene, which encodes for a key enzyme involved in the cellular process of ubiquitylation. The UBA1 gene encodes for the ubiquitin-activating enzyme E1, responsible for the first step of the ubiquitin-proteasome system. This system is responsible for recognizing and targeting proteins that are damaged or no longer needed by the cell, marking them for degradation via the proteasome.
The ubiquitin-activating enzyme E1 is essential for this process, as it catalyzes the covalent attachment of ubiquitin molecules to the proteins to be degraded. This marks them for recognition by the proteasome, allowing it to break them down into their constituent parts. The name VEXAS is derived from the key features of the syndrome, including the presence of vacuoles in myeloid and erythroid progenitor cells, the involvement of the E1 enzyme in the disease, the X-linked inheritance of the UBA1 gene, the autoinflammatory nature of the disease, and the fact that the mutations that cause VEXAS occur somatically in blood cells.
The mutations in UBA1 are specific to myeloid cells and result in chronic inflammation and progressive bone marrow failure, which can be severe and life-threatening. The discovery of VEXAS was made using a genotype-first approach, which revealed that all cases of the disease were caused by new, previously unreported mutations in the UBA1 gene. VEXAS is a severe and advancing disease that displays symptoms that overlap with both rheumatological and hematological disorders.
This disease is characterized by widespread inflammation affecting various body parts, including the skin, lungs, blood vessels, and cartilage. This leads to a range of diagnoses, including sweet syndrome, relapsing polychondritis, polyarteritis nodosa, and giant cell arteritis. Furthermore, patients with VEXAS also experience a range of hematologic issues, including anemia with large red blood cells, low platelet count, an increased risk of blood clots and blockages, and a gradual decline in bone marrow function that can eventually lead to blood cancer.
Epidemiology
The exact prevalence of VEXAS (Vascular Endothelial Growth Factor Inhibitor-Induced VEGF-A Receptor tyrosine kinase inhibitor Syndrome) is unknown. It is a relatively rare and recently recognized side effect of anti-angiogenic cancer therapy. It is used to treat various cancers, including renal cell carcinoma, non-small cell lung cancer, and others.
The syndrome is associated with the use of vascular endothelial growth factor (VEGF) inhibitors, which target the growth of new blood vessels in malignant tumors. The first research conducted suggests that myeloid precursor cells can endure a somatic mutation, while mutant lymphocytes are less likely to survive in the bone marrow.
This is likely due to the toxic effects of the unfolded protein response and proteotoxic stress on lymphocytes compared to myeloid cells. Moreover, the study data indicates that only males are affected by this mutation, which implies that the additional allele seen in female individuals could be a protective factor against the effects of the mutant allele. It is also possible that the disease is less severe for women due to skewed X-inactivation.
Anatomy
Pathophysiology
Ubiquitylation is a three-step process involving the coordinated actions of ubiquitin-activating enzymes (E1), which include two distinct enzymes; ubiquitin-conjugating enzymes (E2), which include nearly 40 distinct enzymes; and substrate-specific ligases (E3), which include over 600 distinct enzymes.
Physiologic modulation of ubiquitin signaling frequently occurs at the E2 and E3 enzymes level, but control of ubiquitin activation is considerably less understood. A crucial role for subcellular controlled ubiquitin activation during hematopoiesis is supported by the VEXAS syndrome, which is a reduction of cytoplasmic UBA1. UBA1 is crucial for almost all cellular ubiquitin signaling in model organisms and cultured cells.
The observation that mutations affecting p. Met41 are fatal when they are germline and that this mutation is only compatible with life when mosaic in particular cell types likely explains why UBA1 variations have only been discovered in the somatic stage. The VEXAS syndrome’s inflammation is fueled by mutant myeloid cells, which outnumber myeloid cells of the wild type.
Etiology
VEXAS syndrome is a complex genetic disorder that is caused by a combination of genetic and environmental factors. The underlying genetic cause of VEXAS is currently unknown, and it is thought to be a multifactorial disorder resulting from the interaction of multiple genes with environmental factors.
Environmental factors that may contribute to the development of VEXAS include exposure to toxins, infections, and maternal health conditions during pregnancy. Further research is needed to fully understand the genetic and environmental factors involved in the development of VEXAS syndrome.
Genetics
Prognostic Factors
The prognosis of VEXAS syndrome is poor and is considered a progressive, degenerative condition. The syndrome typically results in significant neurological decline, loss of mobility, and, eventually, death.
However, the rate of progression and the age of onset can vary between individuals. There is currently no cure for VEXAS syndrome, and treatment is primarily symptomatic and supportive.
Clinical History
Physical Examination
Age group
Associated comorbidity
Associated activity
Acuity of presentation
Differential Diagnoses
Laboratory Studies
Imaging Studies
Procedures
Histologic Findings
Staging
Treatment Paradigm
by Stage
by Modality
Chemotherapy
Radiation Therapy
Surgical Interventions
Hormone Therapy
Immunotherapy
Hyperthermia
Photodynamic Therapy
Stem Cell Transplant
Targeted Therapy
Palliative Care
Medication
Off-label:
1st Treatment Cycle:
75
mg/m²
Subcutaneous (SC) or intravenously (IV)
every day
7
days
Subsequent Treatment Cycles:
Every 4 weeks, repeat the cycles
If no therapeutic impact is shown after two treatment cycles and no harm other than nausea and vomiting has occurred, the dose may be increased to 100 mg/m2
Patients should receive treatment for a minimum of 4 to 6 cycles
However, more treatment cycles might be necessary for a full or partial response
As long as the patient keeps getting well, the treatment may be continued
Off-label:
tocilizumab (162 mg/0.9 mL) weekly injections were started, and daily oral prednisone (40 mg) was maintained
Given that individuals with VEXAS had higher levels of IL-6 and that tocilizumab is a competitive inhibitor of IL-6 binding to its receptor, tocilizumab was chosen as a treatment (IL-6R)
Future Trends
References
VEXAS syndrome is a rare disorder that affects adults and is caused by mutations in the UBA1 gene, which encodes for a key enzyme involved in the cellular process of ubiquitylation. The UBA1 gene encodes for the ubiquitin-activating enzyme E1, responsible for the first step of the ubiquitin-proteasome system. This system is responsible for recognizing and targeting proteins that are damaged or no longer needed by the cell, marking them for degradation via the proteasome.
The ubiquitin-activating enzyme E1 is essential for this process, as it catalyzes the covalent attachment of ubiquitin molecules to the proteins to be degraded. This marks them for recognition by the proteasome, allowing it to break them down into their constituent parts. The name VEXAS is derived from the key features of the syndrome, including the presence of vacuoles in myeloid and erythroid progenitor cells, the involvement of the E1 enzyme in the disease, the X-linked inheritance of the UBA1 gene, the autoinflammatory nature of the disease, and the fact that the mutations that cause VEXAS occur somatically in blood cells.
The mutations in UBA1 are specific to myeloid cells and result in chronic inflammation and progressive bone marrow failure, which can be severe and life-threatening. The discovery of VEXAS was made using a genotype-first approach, which revealed that all cases of the disease were caused by new, previously unreported mutations in the UBA1 gene. VEXAS is a severe and advancing disease that displays symptoms that overlap with both rheumatological and hematological disorders.
This disease is characterized by widespread inflammation affecting various body parts, including the skin, lungs, blood vessels, and cartilage. This leads to a range of diagnoses, including sweet syndrome, relapsing polychondritis, polyarteritis nodosa, and giant cell arteritis. Furthermore, patients with VEXAS also experience a range of hematologic issues, including anemia with large red blood cells, low platelet count, an increased risk of blood clots and blockages, and a gradual decline in bone marrow function that can eventually lead to blood cancer.
The exact prevalence of VEXAS (Vascular Endothelial Growth Factor Inhibitor-Induced VEGF-A Receptor tyrosine kinase inhibitor Syndrome) is unknown. It is a relatively rare and recently recognized side effect of anti-angiogenic cancer therapy. It is used to treat various cancers, including renal cell carcinoma, non-small cell lung cancer, and others.
The syndrome is associated with the use of vascular endothelial growth factor (VEGF) inhibitors, which target the growth of new blood vessels in malignant tumors. The first research conducted suggests that myeloid precursor cells can endure a somatic mutation, while mutant lymphocytes are less likely to survive in the bone marrow.
This is likely due to the toxic effects of the unfolded protein response and proteotoxic stress on lymphocytes compared to myeloid cells. Moreover, the study data indicates that only males are affected by this mutation, which implies that the additional allele seen in female individuals could be a protective factor against the effects of the mutant allele. It is also possible that the disease is less severe for women due to skewed X-inactivation.
Ubiquitylation is a three-step process involving the coordinated actions of ubiquitin-activating enzymes (E1), which include two distinct enzymes; ubiquitin-conjugating enzymes (E2), which include nearly 40 distinct enzymes; and substrate-specific ligases (E3), which include over 600 distinct enzymes.
Physiologic modulation of ubiquitin signaling frequently occurs at the E2 and E3 enzymes level, but control of ubiquitin activation is considerably less understood. A crucial role for subcellular controlled ubiquitin activation during hematopoiesis is supported by the VEXAS syndrome, which is a reduction of cytoplasmic UBA1. UBA1 is crucial for almost all cellular ubiquitin signaling in model organisms and cultured cells.
The observation that mutations affecting p. Met41 are fatal when they are germline and that this mutation is only compatible with life when mosaic in particular cell types likely explains why UBA1 variations have only been discovered in the somatic stage. The VEXAS syndrome’s inflammation is fueled by mutant myeloid cells, which outnumber myeloid cells of the wild type.
VEXAS syndrome is a complex genetic disorder that is caused by a combination of genetic and environmental factors. The underlying genetic cause of VEXAS is currently unknown, and it is thought to be a multifactorial disorder resulting from the interaction of multiple genes with environmental factors.
Environmental factors that may contribute to the development of VEXAS include exposure to toxins, infections, and maternal health conditions during pregnancy. Further research is needed to fully understand the genetic and environmental factors involved in the development of VEXAS syndrome.
The prognosis of VEXAS syndrome is poor and is considered a progressive, degenerative condition. The syndrome typically results in significant neurological decline, loss of mobility, and, eventually, death.
However, the rate of progression and the age of onset can vary between individuals. There is currently no cure for VEXAS syndrome, and treatment is primarily symptomatic and supportive.
VEXAS syndrome is a rare disorder that affects adults and is caused by mutations in the UBA1 gene, which encodes for a key enzyme involved in the cellular process of ubiquitylation. The UBA1 gene encodes for the ubiquitin-activating enzyme E1, responsible for the first step of the ubiquitin-proteasome system. This system is responsible for recognizing and targeting proteins that are damaged or no longer needed by the cell, marking them for degradation via the proteasome.
The ubiquitin-activating enzyme E1 is essential for this process, as it catalyzes the covalent attachment of ubiquitin molecules to the proteins to be degraded. This marks them for recognition by the proteasome, allowing it to break them down into their constituent parts. The name VEXAS is derived from the key features of the syndrome, including the presence of vacuoles in myeloid and erythroid progenitor cells, the involvement of the E1 enzyme in the disease, the X-linked inheritance of the UBA1 gene, the autoinflammatory nature of the disease, and the fact that the mutations that cause VEXAS occur somatically in blood cells.
The mutations in UBA1 are specific to myeloid cells and result in chronic inflammation and progressive bone marrow failure, which can be severe and life-threatening. The discovery of VEXAS was made using a genotype-first approach, which revealed that all cases of the disease were caused by new, previously unreported mutations in the UBA1 gene. VEXAS is a severe and advancing disease that displays symptoms that overlap with both rheumatological and hematological disorders.
This disease is characterized by widespread inflammation affecting various body parts, including the skin, lungs, blood vessels, and cartilage. This leads to a range of diagnoses, including sweet syndrome, relapsing polychondritis, polyarteritis nodosa, and giant cell arteritis. Furthermore, patients with VEXAS also experience a range of hematologic issues, including anemia with large red blood cells, low platelet count, an increased risk of blood clots and blockages, and a gradual decline in bone marrow function that can eventually lead to blood cancer.
The exact prevalence of VEXAS (Vascular Endothelial Growth Factor Inhibitor-Induced VEGF-A Receptor tyrosine kinase inhibitor Syndrome) is unknown. It is a relatively rare and recently recognized side effect of anti-angiogenic cancer therapy. It is used to treat various cancers, including renal cell carcinoma, non-small cell lung cancer, and others.
The syndrome is associated with the use of vascular endothelial growth factor (VEGF) inhibitors, which target the growth of new blood vessels in malignant tumors. The first research conducted suggests that myeloid precursor cells can endure a somatic mutation, while mutant lymphocytes are less likely to survive in the bone marrow.
This is likely due to the toxic effects of the unfolded protein response and proteotoxic stress on lymphocytes compared to myeloid cells. Moreover, the study data indicates that only males are affected by this mutation, which implies that the additional allele seen in female individuals could be a protective factor against the effects of the mutant allele. It is also possible that the disease is less severe for women due to skewed X-inactivation.
Ubiquitylation is a three-step process involving the coordinated actions of ubiquitin-activating enzymes (E1), which include two distinct enzymes; ubiquitin-conjugating enzymes (E2), which include nearly 40 distinct enzymes; and substrate-specific ligases (E3), which include over 600 distinct enzymes.
Physiologic modulation of ubiquitin signaling frequently occurs at the E2 and E3 enzymes level, but control of ubiquitin activation is considerably less understood. A crucial role for subcellular controlled ubiquitin activation during hematopoiesis is supported by the VEXAS syndrome, which is a reduction of cytoplasmic UBA1. UBA1 is crucial for almost all cellular ubiquitin signaling in model organisms and cultured cells.
The observation that mutations affecting p. Met41 are fatal when they are germline and that this mutation is only compatible with life when mosaic in particular cell types likely explains why UBA1 variations have only been discovered in the somatic stage. The VEXAS syndrome’s inflammation is fueled by mutant myeloid cells, which outnumber myeloid cells of the wild type.
VEXAS syndrome is a complex genetic disorder that is caused by a combination of genetic and environmental factors. The underlying genetic cause of VEXAS is currently unknown, and it is thought to be a multifactorial disorder resulting from the interaction of multiple genes with environmental factors.
Environmental factors that may contribute to the development of VEXAS include exposure to toxins, infections, and maternal health conditions during pregnancy. Further research is needed to fully understand the genetic and environmental factors involved in the development of VEXAS syndrome.
The prognosis of VEXAS syndrome is poor and is considered a progressive, degenerative condition. The syndrome typically results in significant neurological decline, loss of mobility, and, eventually, death.
However, the rate of progression and the age of onset can vary between individuals. There is currently no cure for VEXAS syndrome, and treatment is primarily symptomatic and supportive.

Both our subscription plans include Free CME/CPD AMA PRA Category 1 credits.

On course completion, you will receive a full-sized presentation quality digital certificate.
A dynamic medical simulation platform designed to train healthcare professionals and students to effectively run code situations through an immersive hands-on experience in a live, interactive 3D environment.
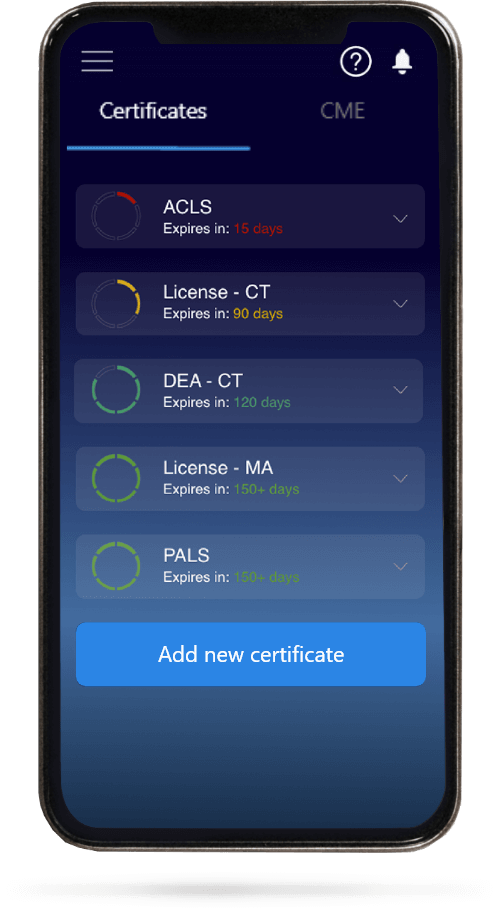
When you have your licenses, certificates and CMEs in one place, it's easier to track your career growth. You can easily share these with hospitals as well, using your medtigo app.