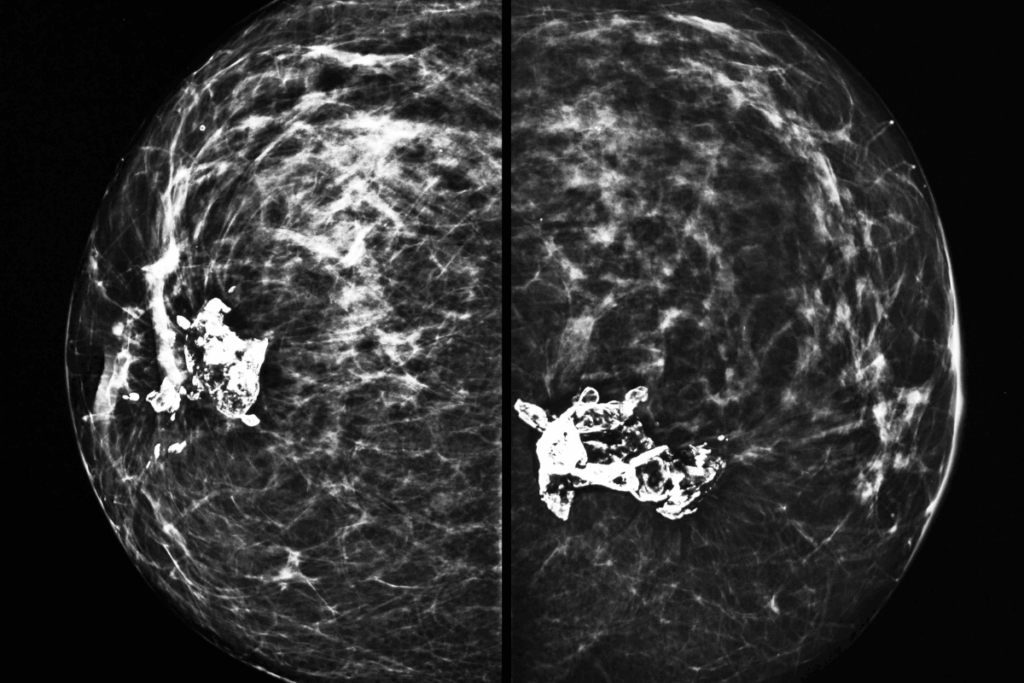
Cardiovascular disease (CVD) is the leading cause of death among women, yet it is often underdiagnosed. Current risk assessment models mainly depend on clinical history, laboratory findings, and traditional risk factors, such as those involved in predicting risk of cardiovascular disease events (PREVENT) calculators. But these models lack direct evaluation of vascular anatomy. Mammography, which is commonly used for breast cancer screening. It offers a visualization of arterial structures in most adult women. Breast arterial calcification (BAC) visible on mammograms has been shown to correlate with calcification in other vascular beds and to predict the future CVD risk. Unlike coronary artery calcification, BAC does not cause luminal obstruction but indicates vascular aging. Traditional BAC assessments have been semi-quantitative or qualitative. A recent study published in European Heart Journal used an advanced automated transformer-based model for the quantification of BAC from mammograms, enabling standardized measurement and improving CVD risk prediction among women.
This retrospective cohort study included a total of 74,124 women (mean age = 55.5±10.4 years) from Emory Healthcare as an internal cohort and 49,638 women (mean age = 59.5±10.2 years) from Mayo Clinic as an external validation cohort. Only one index mammogram per patient was analyzed. Women with prior CVD, major adverse CVD events, and previous coronary imaging were excluded from this study. BAC was quantified by using a transform-based artificial intelligence (AI) model, which was applied to mediolateral oblique mammogram images. BAC severity was classified as severe (n = 714; mean age = 67.8±9.1 years), mild (n = 10,369; mean age = 58.9±10.9 years), none (n = 62,187; mean age = 54.6±10 years), or moderate (n = 854; mean age = 68.4±8.6 years). The median follow-up period was found to be 7 years (interquartile range [IQR]: 4-10). Major adverse cardiovascular events (MACE) and risk factors were detected from electronic health records (EHR). PREVENT risk scores were estimated in comparison with BAC measurements.
BAC was observed in 26% of women in the external cohort and 16.1% in the internal cohort. In the internal cohort, the incidence of MACE increased more than 8-fold from 5.96/1000 person-years in women with zero BAC to 48.89 in those with severe BAC. These event rates were consistently higher in the external cohort. BAC severity was strongly associated with cardiometabolic risk factors and age, with women with severe BAC being about 13 years older compared to those without BAC (p <0.001). Higher systolic blood pressure, diabetes, lower estimated glomerular filtration rate, higher body mass index, and use of statin and antihypertensive were also linked with increasing BAC severity, whereas smoking showed no association.
Severe BAC significantly increased the risk of heart failure in the internal cohort with a hazard ratio (HR) of 2.13 (95% confidence interval [CI]: 1.67-2.73) and all-cause mortality in the external cohort with an HR of 2.00 (95% CI: 1.63-2.45). Each 1mm2 increase in BAC raised the risk by 1 to 2%, and doubling BAC increased MACE risk by 7 to 19%. BAC also improved prediction beyond the PREVENT score, with severe BAC associated with an HR of 2.80 (95% CI: 2.36-3.32) in the external cohort and 3.29 (95% CI: 2.15-5.05) in the internal cohort for MACE. The AI model showed 91% sensitivity and 95% specificity.
This study’s limitations include its retrospective design and use of data from only two US institutions, which may limit generalizability. Potential misclassification from EHR data, missing information on key confounders, limited racial diversity, and modest improvement in predictive performance also warrant caution and need further prospective validation.
In conclusion, this study highlights that automated quantification of BAC from routine mammography may serve as an opportunistic tool for CVD risk assessment among women, supporting early detection without additional radiation and providing predictive value beyond traditional risk factors and PREVENT scores.
Reference: Dapamede T, Urooj A, Joshi V, et al. Artificial intelligence–based quantification of breast arterial calcifications to predict cardiovascular morbidity and mortality. Eur Heart J. 2026:ehag128. doi:10.1093/eurheartj/ehag128











