The epidemiology of A. faecalis reveals its status as a gram-negative bacterium commonly encountered in diverse environments, including soil, water, and healthcare settings. This opportunistic pathogen tends to cause infections in individuals with compromised immunity or underlying health conditions, with a preference for sites such as the bloodstream, urinary tract, skin, & soft tissue, as well as the middle ear.
A retrospective study spanning from 2014 to 2019 underscored concerning trends in antibiotic susceptibility among A. faecalis strains, with an emerging prevalence of extensively drug-resistant (XDR) variants. Notably, the bacterium exhibited decreasing susceptibility to commonly employed antibiotics during this period. In 2019, the highest sensitivity rate was observed for three antibiotics (imipenem, meropenem, and ceftazidime), reaching 66.7%, while ciprofloxacin and piperacillin/tazobactam displayed sensitivity rates below 50%.
Geographical variations in A. faecalis incidence were evident, as illustrated by its isolation from diverse samples. In a hospital in India from 2010 to 2012, A. faecalis was identified in 0.3% of urine samples and 0.2% of wound samples. Similarly, from 2011 to 2012, the bacterium was detected in 0.4% of middle ear fluid samples from children with otitis media in Korea.
Classification and Structure:
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Betaproteobacteria
Order: Burkholderiales
Family: Alcaligenaceae
Genus: Alcaligenes
Species: A. faecalis
Alcaligenes faecalis is characterized by its gram-negative nature, exhibiting a rod-shaped or coccal-rod-shaped morphology. The bacterium is equipped with peritrichous flagella, providing it with the ability to move in various directions.
While it can form capsules, it does not produce spores as part of its reproductive strategy. The typical dimensions of A. faecalis cells range from approximately 0.5 to 1.0 μm in width and 0.5 to 2.6 μm in length.
A. faecalis possesses a repertoire of virulence and antimicrobial resistance-related proteins, including hemolysin, lipase, protease, siderophore, oxidase, catalase, nitrate reductase, and efflux pumps.
These proteins collectively contribute to various aspects of bacterial survival and pathogenicity, enabling the bacterium to lyse host cells, degrade host lipids and proteins, acquire essential nutrients like iron, detoxify reactive oxygen species, and counteract antimicrobial compounds. Additionally, A. faecalis produces diverse β-lactamases capable of hydrolyzing various penicillins and cephalosporins.
The type strain of Alcaligenes faecalis, designated as 16 (R. Hugh 135), holds cultural significance with additional identifiers such as DSM 30030 and ATCC 8750. Isolated initially from feces by H.J. Conn, this strain has been a subject of scientific exploration since its description by Castellani and Chalmers in 1919. The genetic similarities shared by A. faecalis with other bacteria, such as A. eutrophus and Bordetella pertussis, contribute to its evolutionary relationships and functional characteristics within microbial ecosystems.
The pathogenesis of Alcaligenes faecalis in humans remains a subject of limited understanding, yet several potential mechanisms shed light on its interactions with the host. A. faecalis exhibits the capacity to adhere to and invade diverse human cells, including epithelial and endothelial cells.
Additionally, its ability to form biofilms on medical devices and surfaces enhances colonization and persistence in host tissues. The production of various toxins, such as hemolysin, lipase, protease, and siderophore, contributes to the host cell and tissue damage, aiding in the acquisition of essential nutrients like iron.
A. faecalis employs strategies to evade the host immune system, including capsule production, antigenic variation, and modulation of cytokine production. These tactics allow the bacterium to resist clearance by the immune response.
Furthermore, A. faecalis demonstrates resistance to a range of antibiotics, encompassing penicillins, cephalosporins, aminoglycosides, quinolones, and carbapenems. The acquisition of resistance genes via horizontal gene transfer from other bacteria further complicates treatment, rendering the bacterium challenging to eradicate and elevating the risk of treatment failure and associated complications.
The immune response against A. faecalis involves a coordinated interplay between various components of the immune system. Upon exposure to the bacterium, B cells may be directly activated or indirectly through helper T cells and dendritic cells, leading to the production of antibodies. A. faecalis can be recognized by pattern recognition receptors (PRRs), and phagocytosis is facilitated by opsonization, where the bacterium is coated with antibodies or complement proteins, aiding its uptake by phagocytes.
Inflammation is a crucial mechanism that helps recruit and activate immune cells, increase blood flow, enhance permeability, and facilitate the delivery of antimicrobial agents to the infection site. However, it’s important to note that excessive or chronic inflammation can also lead to tissue damage & systemic complications.
Alcaligenes faecalis is capable of triggering inflammation by stimulating specific PRRs, including Toll-like receptors (TLR2, TLR4) and NOD-like receptor family pyrin domain-containing 3 (NLRP3), on immune and other cells. Additionally, the bacterium can induce nitric oxide (NO) production via the activation of PRRs, such as TLR4 and NLRP3, on immune cells. It’s noteworthy that certain strains of A. faecalis have evolved mechanisms to evade NO killing, either by producing low levels of lipopolysaccharide or by expressing efflux pumps that expel NO.
A. faecalis infection can manifest in various clinical presentations, with bacteremia being a prominent feature. Bacteremia occurs when the Alcaligenes enter the bloodstream, leading to systemic symptoms such as fever, chills, and low blood pressure. This condition poses a risk of disseminating the infection to different organs and tissues throughout the body.
Another common manifestation is urinary tract infection (UTI), where A. faecalis infects the urinary system. Patients with A. faecalis UTI may experience symptoms like frequent urges to urinate, a burning sensation during urination, & the presence of cloudy or bloody urine.
Skin and soft tissue infections represent another clinical aspect of A. faecalis infection. In these cases, the bacteria invade the skin or underlying tissues, resulting in local symptoms like redness, swelling, pain, & the formation of pus. These infections may occur through wounds, breaks in the skin, or other entry points.
In more severe cases, A. faecalis can cause meningitis by infecting the membranes covering the brain and spinal cord. This condition presents with symptoms including headaches, stiff neck, and tachypnea (fast, shallow breathing). Meningitis represents a severe and potentially life-threatening complication associated with A. faecalis infection.
The epidemiology of A. faecalis reveals its status as a gram-negative bacterium commonly encountered in diverse environments, including soil, water, and healthcare settings. This opportunistic pathogen tends to cause infections in individuals with compromised immunity or underlying health conditions, with a preference for sites such as the bloodstream, urinary tract, skin, & soft tissue, as well as the middle ear.
A retrospective study spanning from 2014 to 2019 underscored concerning trends in antibiotic susceptibility among A. faecalis strains, with an emerging prevalence of extensively drug-resistant (XDR) variants. Notably, the bacterium exhibited decreasing susceptibility to commonly employed antibiotics during this period. In 2019, the highest sensitivity rate was observed for three antibiotics (imipenem, meropenem, and ceftazidime), reaching 66.7%, while ciprofloxacin and piperacillin/tazobactam displayed sensitivity rates below 50%.
Geographical variations in A. faecalis incidence were evident, as illustrated by its isolation from diverse samples. In a hospital in India from 2010 to 2012, A. faecalis was identified in 0.3% of urine samples and 0.2% of wound samples. Similarly, from 2011 to 2012, the bacterium was detected in 0.4% of middle ear fluid samples from children with otitis media in Korea.
Classification and Structure:
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Betaproteobacteria
Order: Burkholderiales
Family: Alcaligenaceae
Genus: Alcaligenes
Species: A. faecalis
Alcaligenes faecalis is characterized by its gram-negative nature, exhibiting a rod-shaped or coccal-rod-shaped morphology. The bacterium is equipped with peritrichous flagella, providing it with the ability to move in various directions.
While it can form capsules, it does not produce spores as part of its reproductive strategy. The typical dimensions of A. faecalis cells range from approximately 0.5 to 1.0 μm in width and 0.5 to 2.6 μm in length.
A. faecalis possesses a repertoire of virulence and antimicrobial resistance-related proteins, including hemolysin, lipase, protease, siderophore, oxidase, catalase, nitrate reductase, and efflux pumps.
These proteins collectively contribute to various aspects of bacterial survival and pathogenicity, enabling the bacterium to lyse host cells, degrade host lipids and proteins, acquire essential nutrients like iron, detoxify reactive oxygen species, and counteract antimicrobial compounds. Additionally, A. faecalis produces diverse β-lactamases capable of hydrolyzing various penicillins and cephalosporins.
The type strain of Alcaligenes faecalis, designated as 16 (R. Hugh 135), holds cultural significance with additional identifiers such as DSM 30030 and ATCC 8750. Isolated initially from feces by H.J. Conn, this strain has been a subject of scientific exploration since its description by Castellani and Chalmers in 1919. The genetic similarities shared by A. faecalis with other bacteria, such as A. eutrophus and Bordetella pertussis, contribute to its evolutionary relationships and functional characteristics within microbial ecosystems.
The pathogenesis of Alcaligenes faecalis in humans remains a subject of limited understanding, yet several potential mechanisms shed light on its interactions with the host. A. faecalis exhibits the capacity to adhere to and invade diverse human cells, including epithelial and endothelial cells.
Additionally, its ability to form biofilms on medical devices and surfaces enhances colonization and persistence in host tissues. The production of various toxins, such as hemolysin, lipase, protease, and siderophore, contributes to the host cell and tissue damage, aiding in the acquisition of essential nutrients like iron.
A. faecalis employs strategies to evade the host immune system, including capsule production, antigenic variation, and modulation of cytokine production. These tactics allow the bacterium to resist clearance by the immune response.
Furthermore, A. faecalis demonstrates resistance to a range of antibiotics, encompassing penicillins, cephalosporins, aminoglycosides, quinolones, and carbapenems. The acquisition of resistance genes via horizontal gene transfer from other bacteria further complicates treatment, rendering the bacterium challenging to eradicate and elevating the risk of treatment failure and associated complications.
The immune response against A. faecalis involves a coordinated interplay between various components of the immune system. Upon exposure to the bacterium, B cells may be directly activated or indirectly through helper T cells and dendritic cells, leading to the production of antibodies. A. faecalis can be recognized by pattern recognition receptors (PRRs), and phagocytosis is facilitated by opsonization, where the bacterium is coated with antibodies or complement proteins, aiding its uptake by phagocytes.
Inflammation is a crucial mechanism that helps recruit and activate immune cells, increase blood flow, enhance permeability, and facilitate the delivery of antimicrobial agents to the infection site. However, it’s important to note that excessive or chronic inflammation can also lead to tissue damage & systemic complications.
Alcaligenes faecalis is capable of triggering inflammation by stimulating specific PRRs, including Toll-like receptors (TLR2, TLR4) and NOD-like receptor family pyrin domain-containing 3 (NLRP3), on immune and other cells. Additionally, the bacterium can induce nitric oxide (NO) production via the activation of PRRs, such as TLR4 and NLRP3, on immune cells. It’s noteworthy that certain strains of A. faecalis have evolved mechanisms to evade NO killing, either by producing low levels of lipopolysaccharide or by expressing efflux pumps that expel NO.
A. faecalis infection can manifest in various clinical presentations, with bacteremia being a prominent feature. Bacteremia occurs when the Alcaligenes enter the bloodstream, leading to systemic symptoms such as fever, chills, and low blood pressure. This condition poses a risk of disseminating the infection to different organs and tissues throughout the body.
Another common manifestation is urinary tract infection (UTI), where A. faecalis infects the urinary system. Patients with A. faecalis UTI may experience symptoms like frequent urges to urinate, a burning sensation during urination, & the presence of cloudy or bloody urine.
Skin and soft tissue infections represent another clinical aspect of A. faecalis infection. In these cases, the bacteria invade the skin or underlying tissues, resulting in local symptoms like redness, swelling, pain, & the formation of pus. These infections may occur through wounds, breaks in the skin, or other entry points.
In more severe cases, A. faecalis can cause meningitis by infecting the membranes covering the brain and spinal cord. This condition presents with symptoms including headaches, stiff neck, and tachypnea (fast, shallow breathing). Meningitis represents a severe and potentially life-threatening complication associated with A. faecalis infection.
The epidemiology of A. faecalis reveals its status as a gram-negative bacterium commonly encountered in diverse environments, including soil, water, and healthcare settings. This opportunistic pathogen tends to cause infections in individuals with compromised immunity or underlying health conditions, with a preference for sites such as the bloodstream, urinary tract, skin, & soft tissue, as well as the middle ear.
A retrospective study spanning from 2014 to 2019 underscored concerning trends in antibiotic susceptibility among A. faecalis strains, with an emerging prevalence of extensively drug-resistant (XDR) variants. Notably, the bacterium exhibited decreasing susceptibility to commonly employed antibiotics during this period. In 2019, the highest sensitivity rate was observed for three antibiotics (imipenem, meropenem, and ceftazidime), reaching 66.7%, while ciprofloxacin and piperacillin/tazobactam displayed sensitivity rates below 50%.
Geographical variations in A. faecalis incidence were evident, as illustrated by its isolation from diverse samples. In a hospital in India from 2010 to 2012, A. faecalis was identified in 0.3% of urine samples and 0.2% of wound samples. Similarly, from 2011 to 2012, the bacterium was detected in 0.4% of middle ear fluid samples from children with otitis media in Korea.
Classification and Structure:
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Betaproteobacteria
Order: Burkholderiales
Family: Alcaligenaceae
Genus: Alcaligenes
Species: A. faecalis
Alcaligenes faecalis is characterized by its gram-negative nature, exhibiting a rod-shaped or coccal-rod-shaped morphology. The bacterium is equipped with peritrichous flagella, providing it with the ability to move in various directions.
While it can form capsules, it does not produce spores as part of its reproductive strategy. The typical dimensions of A. faecalis cells range from approximately 0.5 to 1.0 μm in width and 0.5 to 2.6 μm in length.
A. faecalis possesses a repertoire of virulence and antimicrobial resistance-related proteins, including hemolysin, lipase, protease, siderophore, oxidase, catalase, nitrate reductase, and efflux pumps.
These proteins collectively contribute to various aspects of bacterial survival and pathogenicity, enabling the bacterium to lyse host cells, degrade host lipids and proteins, acquire essential nutrients like iron, detoxify reactive oxygen species, and counteract antimicrobial compounds. Additionally, A. faecalis produces diverse β-lactamases capable of hydrolyzing various penicillins and cephalosporins.
The type strain of Alcaligenes faecalis, designated as 16 (R. Hugh 135), holds cultural significance with additional identifiers such as DSM 30030 and ATCC 8750. Isolated initially from feces by H.J. Conn, this strain has been a subject of scientific exploration since its description by Castellani and Chalmers in 1919. The genetic similarities shared by A. faecalis with other bacteria, such as A. eutrophus and Bordetella pertussis, contribute to its evolutionary relationships and functional characteristics within microbial ecosystems.
The pathogenesis of Alcaligenes faecalis in humans remains a subject of limited understanding, yet several potential mechanisms shed light on its interactions with the host. A. faecalis exhibits the capacity to adhere to and invade diverse human cells, including epithelial and endothelial cells.
Additionally, its ability to form biofilms on medical devices and surfaces enhances colonization and persistence in host tissues. The production of various toxins, such as hemolysin, lipase, protease, and siderophore, contributes to the host cell and tissue damage, aiding in the acquisition of essential nutrients like iron.
A. faecalis employs strategies to evade the host immune system, including capsule production, antigenic variation, and modulation of cytokine production. These tactics allow the bacterium to resist clearance by the immune response.
Furthermore, A. faecalis demonstrates resistance to a range of antibiotics, encompassing penicillins, cephalosporins, aminoglycosides, quinolones, and carbapenems. The acquisition of resistance genes via horizontal gene transfer from other bacteria further complicates treatment, rendering the bacterium challenging to eradicate and elevating the risk of treatment failure and associated complications.
The immune response against A. faecalis involves a coordinated interplay between various components of the immune system. Upon exposure to the bacterium, B cells may be directly activated or indirectly through helper T cells and dendritic cells, leading to the production of antibodies. A. faecalis can be recognized by pattern recognition receptors (PRRs), and phagocytosis is facilitated by opsonization, where the bacterium is coated with antibodies or complement proteins, aiding its uptake by phagocytes.
Inflammation is a crucial mechanism that helps recruit and activate immune cells, increase blood flow, enhance permeability, and facilitate the delivery of antimicrobial agents to the infection site. However, it’s important to note that excessive or chronic inflammation can also lead to tissue damage & systemic complications.
Alcaligenes faecalis is capable of triggering inflammation by stimulating specific PRRs, including Toll-like receptors (TLR2, TLR4) and NOD-like receptor family pyrin domain-containing 3 (NLRP3), on immune and other cells. Additionally, the bacterium can induce nitric oxide (NO) production via the activation of PRRs, such as TLR4 and NLRP3, on immune cells. It’s noteworthy that certain strains of A. faecalis have evolved mechanisms to evade NO killing, either by producing low levels of lipopolysaccharide or by expressing efflux pumps that expel NO.
A. faecalis infection can manifest in various clinical presentations, with bacteremia being a prominent feature. Bacteremia occurs when the Alcaligenes enter the bloodstream, leading to systemic symptoms such as fever, chills, and low blood pressure. This condition poses a risk of disseminating the infection to different organs and tissues throughout the body.
Another common manifestation is urinary tract infection (UTI), where A. faecalis infects the urinary system. Patients with A. faecalis UTI may experience symptoms like frequent urges to urinate, a burning sensation during urination, & the presence of cloudy or bloody urine.
Skin and soft tissue infections represent another clinical aspect of A. faecalis infection. In these cases, the bacteria invade the skin or underlying tissues, resulting in local symptoms like redness, swelling, pain, & the formation of pus. These infections may occur through wounds, breaks in the skin, or other entry points.
In more severe cases, A. faecalis can cause meningitis by infecting the membranes covering the brain and spinal cord. This condition presents with symptoms including headaches, stiff neck, and tachypnea (fast, shallow breathing). Meningitis represents a severe and potentially life-threatening complication associated with A. faecalis infection.

Both our subscription plans include Free CME/CPD AMA PRA Category 1 credits.

On course completion, you will receive a full-sized presentation quality digital certificate.
A dynamic medical simulation platform designed to train healthcare professionals and students to effectively run code situations through an immersive hands-on experience in a live, interactive 3D environment.
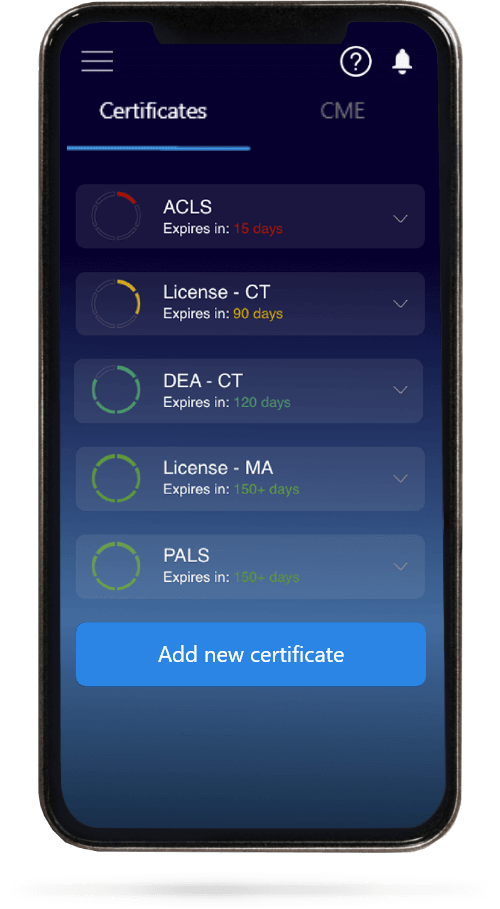
When you have your licenses, certificates and CMEs in one place, it's easier to track your career growth. You can easily share these with hospitals as well, using your medtigo app.