Fasciolopsis buski, a parasitic worm causing intestinal infection, is contracted by consuming aquatic plants contaminated with its larvae. These plants, including water chestnuts and water caltrops, serve as a vehicle for transmission. The geographical distribution of this worm is primarily observed in some areas of Asia, particularly China, India, Thailand, Vietnam, and Bangladesh. A few infections have also been identified in parts of Africa and South America.
The prevalence of Fasciolopsis buski infection exhibits variations influenced by factors such as seasonality and location. In rural areas with close proximity to water bodies and animals acting as reservoir hosts—like pigs and dogs—the infection tends to be more prevalent. The onset of the rainy season, fostering increased aquatic plant growth and snail activity, can lead to higher infection rates. This ailment also exhibits distinct preferences regarding age and gender, with children and females, who may consume raw or undercooked aquatic plants, being more susceptible.
The quantification of global fasciolopsiasis cases is challenging due to the infection’s often asymptomatic nature and potential misdiagnosis. Nevertheless, estimates point towards around 10 million individuals affected worldwide, a substantial portion residing in China, with approximately 2.4 million cases. Regions like India have reported exceptionally high prevalence rates, reaching up to 80% infection rates in certain villages. Also, outbreaks have emerged in Thailand, Vietnam, and Bangladesh.
Classification and Structure:
Kingdom: Animalia
Phylum: Platyhelminthes
Class: Trematoda
Order: Plagiorchiida
Family: Fasciolidae
Genus: Fasciolopsis
Species: Fasciolopsis buski
Fasciolopsis buski is recognized as the largest intestinal fluke in humans, with dimensions ranging from approximately 2 cm to a maximum of 7.5 cm in length and 2.5 cm in width. Its body is elongated and ovoidal, featuring a blunt anterior end.
The fluke possesses two suckers—a larger ventral sucker and a smaller oral sucker—facilitating attachment to the host’s intestinal wall. These suckers aid in anchoring the parasite securely. Fasciolopsis buski exhibits a simple digestive system comprising a mouth, a pharynx, and unbranched ceca, sac-like cavities featuring single openings.
Its reproductive system is more intricate, with tandem dendritic testes, branched ovaries, and extensive vitelline follicles responsible for eggshell production. Unlike other fasciolids, this fluke lacks a cephalic cone or “shoulders.”
The somatic antigens of Fasciolopsis buski are crucial in triggering immune responses within the host. A prime example is the 66 kDa protein antigen, which has been demonstrated to induce the production of IgG and IgE antibodies in individuals infected with the parasite. These antigens stimulate the host’s immune system to mount robust antibody or cellular responses.
Excretory-secretory antigens constitute another essential facet of F. buski‘s interaction with its host’s immune system. These antigens can stimulate the production of antibodies or trigger cellular immune responses within the host. Mitochondrial genes are a pivotal genetic component of F. buski, encoding proteins integral to the parasite’s mitochondrial functions.
These genes encompass 12 protein-coding genes (PCGs), 22 tRNA genes, and two rRNA genes. It might be implicated in crucial processes such as energy metabolism, oxidative stress responses, and drug resistance. Also, the cytochrome c oxidase subunit-1 (cox1) gene of F. buski is a significant molecular marker employed in studies related to the parasite’s phylogenetic relationships and population genetics.
Infection with Fasciolopsis buski occurs when humans ingest contaminated water or aquatic plants containing infectious metacercariae—encysted parasite larvae. Upon ingestion, these larvae excyst within the duodenum and attach themselves to the intestinal surfaces using suckers. Over about three months, these metacercariae develop into adult worms, growing to an impressive size of 7.5 cm by 2.5 cm, making them the largest intestinal flukes known to infect humans.
These mature worms establish their residence in the proximal small bowel and produce eggs expelled through the feces. The impact of F. buski infection on the host’s health is multifaceted. The adult worms provoke harm to the intestinal mucosa through a combination of traumatic, mechanical, and toxic effects. Their presence sparks inflammation and ulceration of the mucosal lining, leading to complications such as bleeding, malabsorption, and diarrhea.
In more severe cases, the worms can instigate partial or complete bowel obstruction, resulting in intense abdominal discomfort, vomiting, nausea, and fever. Beyond the confines of the intestines, these worms may also migrate to other organs like the lungs or liver, giving rise to ectopic lesions. Remarkably, each day, the adult worms generate over 25,000 eggs.
The lifecycle of F. buski encompasses various stages, from the discharge of immature, unembryonated eggs into the intestine and stool. Over weeks, these eggs mature and hatch in water, giving rise to miracidia—tiny parasitic entities—that infiltrate a suitable snail intermediate host. Within the snail, the parasite undergoes a sequence of developmental transformations (sporocysts, rediae, and cercariae).
Subsequently, the cercariae are liberated by the snail and encyst as metacercariae on aquatic plants. When humans or other appropriate final hosts consume these plants, the metacercariae excyst within the duodenum over approximately three months and firmly attach themselves to the intestinal wall.
The consequences of F. buski infection can be wide-ranging. Bile duct blockage can occur, leading to infections or cholangitis, with fever, abdominal pain, and jaundice. Impaired nutrient absorption can result in malabsorption syndrome, accompanied by protein loss, edema, and ascites. Moreover, the worms can instigate hypersensitivity reactions in individuals, triggering responses like urticaria, angioedema, bronchospasm, or even anaphylaxis.
Fasciolopsis buski triggers a range of immune responses in the human host. The innate immune system provides initial defense. Intestinal epithelial cells form a barrier against invasion, aided by mucus that traps and obstructs parasites. Gastric acids and enzymes in the stomach can degrade ingested pathogens. Additionally, immune cells like macrophages and neutrophils perform phagocytosis of the parasite larvae, countering its establishment.
The adaptive immune response offers specific, targeted defense. T lymphocytes, including helper T cells (Th cells) and cytotoxic T cells (Tc cells), are pivotal. Th cells activate other immune cells and boost their efficacy, whereas Tc cells directly eliminate infected cells. B lymphocytes produce antibodies that neutralize the parasite, hindering its attachment and penetration. Cytokines like interferon-gamma (IFN-γ), generated by Th1 cells, enhance macrophage activity, further curbing the infection.
Fasciolopsis buski, recognized as the largest intestinal fluke afflicting humans, is responsible for causing fasciolopsiasis. The clinical expressions of fasciolopsiasis exhibit a spectrum contingent upon the intensity and duration of the infection. While some individuals may remain asymptomatic or experience only mild symptoms such as abdominal pain and diarrhea, instances of more substantial infestations can lead to greater complications.
In instances of heavy infection, these worms can precipitate severe medical concerns. Firstly, intestinal obstruction may arise, inciting intense abdominal pain, vomiting, nausea, and fever. Furthermore, the obstruction of bile ducts—critical conduits for bile transportation—can usher in complications like bile duct infections or cholangitis, characterized by symptoms such as fever, jaundice, and abdominal discomfort.
Moreover, the interference with nutrient absorption—known as malabsorption—can foster protein loss, resulting in the development of edema and ascites. Anemia may ensue due to blood loss from the intestinal wall, further exacerbating the spectrum of clinical implications. In specific scenarios, ectopic lesions might materialize, locating themselves in the intestinal wall, lungs, or other organs, extending the potential complications of this infection.
Diagnosing Fasciolopsis buski infection primarily involves identifying the parasite’s eggs or adult worms within stool samples, duodenal contents, or biliary material. The eggs of F. buski are notable for their size and oval shape, measuring between 130 to 150 μm in length and 60 to 90 μm in width. It’s important to differentiate these eggs from those of Fasciola hepatica, another liver fluke, utilizing alternative methods.
Various techniques are employed to detect F. buski eggs in stool samples:
Additionally, there are instances where the adult worms of F. buski can be located in stool samples or vomitus from infected individuals. In the diagnostic process, specific blood tests can provide valuable insights, including:
Routine blood analysis might reveal eosinophilia—an elevation in white blood cells associated with parasitic infections. Anemia—a reduction in red blood cells—and hypoalbuminemia—a decrease in a specific blood protein—might also be observable.
Fasciolopsis buski, a parasitic worm causing intestinal infection, is contracted by consuming aquatic plants contaminated with its larvae. These plants, including water chestnuts and water caltrops, serve as a vehicle for transmission. The geographical distribution of this worm is primarily observed in some areas of Asia, particularly China, India, Thailand, Vietnam, and Bangladesh. A few infections have also been identified in parts of Africa and South America.
The prevalence of Fasciolopsis buski infection exhibits variations influenced by factors such as seasonality and location. In rural areas with close proximity to water bodies and animals acting as reservoir hosts—like pigs and dogs—the infection tends to be more prevalent. The onset of the rainy season, fostering increased aquatic plant growth and snail activity, can lead to higher infection rates. This ailment also exhibits distinct preferences regarding age and gender, with children and females, who may consume raw or undercooked aquatic plants, being more susceptible.
The quantification of global fasciolopsiasis cases is challenging due to the infection’s often asymptomatic nature and potential misdiagnosis. Nevertheless, estimates point towards around 10 million individuals affected worldwide, a substantial portion residing in China, with approximately 2.4 million cases. Regions like India have reported exceptionally high prevalence rates, reaching up to 80% infection rates in certain villages. Also, outbreaks have emerged in Thailand, Vietnam, and Bangladesh.
Classification and Structure:
Kingdom: Animalia
Phylum: Platyhelminthes
Class: Trematoda
Order: Plagiorchiida
Family: Fasciolidae
Genus: Fasciolopsis
Species: Fasciolopsis buski
Fasciolopsis buski is recognized as the largest intestinal fluke in humans, with dimensions ranging from approximately 2 cm to a maximum of 7.5 cm in length and 2.5 cm in width. Its body is elongated and ovoidal, featuring a blunt anterior end.
The fluke possesses two suckers—a larger ventral sucker and a smaller oral sucker—facilitating attachment to the host’s intestinal wall. These suckers aid in anchoring the parasite securely. Fasciolopsis buski exhibits a simple digestive system comprising a mouth, a pharynx, and unbranched ceca, sac-like cavities featuring single openings.
Its reproductive system is more intricate, with tandem dendritic testes, branched ovaries, and extensive vitelline follicles responsible for eggshell production. Unlike other fasciolids, this fluke lacks a cephalic cone or “shoulders.”
The somatic antigens of Fasciolopsis buski are crucial in triggering immune responses within the host. A prime example is the 66 kDa protein antigen, which has been demonstrated to induce the production of IgG and IgE antibodies in individuals infected with the parasite. These antigens stimulate the host’s immune system to mount robust antibody or cellular responses.
Excretory-secretory antigens constitute another essential facet of F. buski‘s interaction with its host’s immune system. These antigens can stimulate the production of antibodies or trigger cellular immune responses within the host. Mitochondrial genes are a pivotal genetic component of F. buski, encoding proteins integral to the parasite’s mitochondrial functions.
These genes encompass 12 protein-coding genes (PCGs), 22 tRNA genes, and two rRNA genes. It might be implicated in crucial processes such as energy metabolism, oxidative stress responses, and drug resistance. Also, the cytochrome c oxidase subunit-1 (cox1) gene of F. buski is a significant molecular marker employed in studies related to the parasite’s phylogenetic relationships and population genetics.
Infection with Fasciolopsis buski occurs when humans ingest contaminated water or aquatic plants containing infectious metacercariae—encysted parasite larvae. Upon ingestion, these larvae excyst within the duodenum and attach themselves to the intestinal surfaces using suckers. Over about three months, these metacercariae develop into adult worms, growing to an impressive size of 7.5 cm by 2.5 cm, making them the largest intestinal flukes known to infect humans.
These mature worms establish their residence in the proximal small bowel and produce eggs expelled through the feces. The impact of F. buski infection on the host’s health is multifaceted. The adult worms provoke harm to the intestinal mucosa through a combination of traumatic, mechanical, and toxic effects. Their presence sparks inflammation and ulceration of the mucosal lining, leading to complications such as bleeding, malabsorption, and diarrhea.
In more severe cases, the worms can instigate partial or complete bowel obstruction, resulting in intense abdominal discomfort, vomiting, nausea, and fever. Beyond the confines of the intestines, these worms may also migrate to other organs like the lungs or liver, giving rise to ectopic lesions. Remarkably, each day, the adult worms generate over 25,000 eggs.
The lifecycle of F. buski encompasses various stages, from the discharge of immature, unembryonated eggs into the intestine and stool. Over weeks, these eggs mature and hatch in water, giving rise to miracidia—tiny parasitic entities—that infiltrate a suitable snail intermediate host. Within the snail, the parasite undergoes a sequence of developmental transformations (sporocysts, rediae, and cercariae).
Subsequently, the cercariae are liberated by the snail and encyst as metacercariae on aquatic plants. When humans or other appropriate final hosts consume these plants, the metacercariae excyst within the duodenum over approximately three months and firmly attach themselves to the intestinal wall.
The consequences of F. buski infection can be wide-ranging. Bile duct blockage can occur, leading to infections or cholangitis, with fever, abdominal pain, and jaundice. Impaired nutrient absorption can result in malabsorption syndrome, accompanied by protein loss, edema, and ascites. Moreover, the worms can instigate hypersensitivity reactions in individuals, triggering responses like urticaria, angioedema, bronchospasm, or even anaphylaxis.
Fasciolopsis buski triggers a range of immune responses in the human host. The innate immune system provides initial defense. Intestinal epithelial cells form a barrier against invasion, aided by mucus that traps and obstructs parasites. Gastric acids and enzymes in the stomach can degrade ingested pathogens. Additionally, immune cells like macrophages and neutrophils perform phagocytosis of the parasite larvae, countering its establishment.
The adaptive immune response offers specific, targeted defense. T lymphocytes, including helper T cells (Th cells) and cytotoxic T cells (Tc cells), are pivotal. Th cells activate other immune cells and boost their efficacy, whereas Tc cells directly eliminate infected cells. B lymphocytes produce antibodies that neutralize the parasite, hindering its attachment and penetration. Cytokines like interferon-gamma (IFN-γ), generated by Th1 cells, enhance macrophage activity, further curbing the infection.
Fasciolopsis buski, recognized as the largest intestinal fluke afflicting humans, is responsible for causing fasciolopsiasis. The clinical expressions of fasciolopsiasis exhibit a spectrum contingent upon the intensity and duration of the infection. While some individuals may remain asymptomatic or experience only mild symptoms such as abdominal pain and diarrhea, instances of more substantial infestations can lead to greater complications.
In instances of heavy infection, these worms can precipitate severe medical concerns. Firstly, intestinal obstruction may arise, inciting intense abdominal pain, vomiting, nausea, and fever. Furthermore, the obstruction of bile ducts—critical conduits for bile transportation—can usher in complications like bile duct infections or cholangitis, characterized by symptoms such as fever, jaundice, and abdominal discomfort.
Moreover, the interference with nutrient absorption—known as malabsorption—can foster protein loss, resulting in the development of edema and ascites. Anemia may ensue due to blood loss from the intestinal wall, further exacerbating the spectrum of clinical implications. In specific scenarios, ectopic lesions might materialize, locating themselves in the intestinal wall, lungs, or other organs, extending the potential complications of this infection.
Diagnosing Fasciolopsis buski infection primarily involves identifying the parasite’s eggs or adult worms within stool samples, duodenal contents, or biliary material. The eggs of F. buski are notable for their size and oval shape, measuring between 130 to 150 μm in length and 60 to 90 μm in width. It’s important to differentiate these eggs from those of Fasciola hepatica, another liver fluke, utilizing alternative methods.
Various techniques are employed to detect F. buski eggs in stool samples:
Additionally, there are instances where the adult worms of F. buski can be located in stool samples or vomitus from infected individuals. In the diagnostic process, specific blood tests can provide valuable insights, including:
Routine blood analysis might reveal eosinophilia—an elevation in white blood cells associated with parasitic infections. Anemia—a reduction in red blood cells—and hypoalbuminemia—a decrease in a specific blood protein—might also be observable.
Fasciolopsis buski, a parasitic worm causing intestinal infection, is contracted by consuming aquatic plants contaminated with its larvae. These plants, including water chestnuts and water caltrops, serve as a vehicle for transmission. The geographical distribution of this worm is primarily observed in some areas of Asia, particularly China, India, Thailand, Vietnam, and Bangladesh. A few infections have also been identified in parts of Africa and South America.
The prevalence of Fasciolopsis buski infection exhibits variations influenced by factors such as seasonality and location. In rural areas with close proximity to water bodies and animals acting as reservoir hosts—like pigs and dogs—the infection tends to be more prevalent. The onset of the rainy season, fostering increased aquatic plant growth and snail activity, can lead to higher infection rates. This ailment also exhibits distinct preferences regarding age and gender, with children and females, who may consume raw or undercooked aquatic plants, being more susceptible.
The quantification of global fasciolopsiasis cases is challenging due to the infection’s often asymptomatic nature and potential misdiagnosis. Nevertheless, estimates point towards around 10 million individuals affected worldwide, a substantial portion residing in China, with approximately 2.4 million cases. Regions like India have reported exceptionally high prevalence rates, reaching up to 80% infection rates in certain villages. Also, outbreaks have emerged in Thailand, Vietnam, and Bangladesh.
Classification and Structure:
Kingdom: Animalia
Phylum: Platyhelminthes
Class: Trematoda
Order: Plagiorchiida
Family: Fasciolidae
Genus: Fasciolopsis
Species: Fasciolopsis buski
Fasciolopsis buski is recognized as the largest intestinal fluke in humans, with dimensions ranging from approximately 2 cm to a maximum of 7.5 cm in length and 2.5 cm in width. Its body is elongated and ovoidal, featuring a blunt anterior end.
The fluke possesses two suckers—a larger ventral sucker and a smaller oral sucker—facilitating attachment to the host’s intestinal wall. These suckers aid in anchoring the parasite securely. Fasciolopsis buski exhibits a simple digestive system comprising a mouth, a pharynx, and unbranched ceca, sac-like cavities featuring single openings.
Its reproductive system is more intricate, with tandem dendritic testes, branched ovaries, and extensive vitelline follicles responsible for eggshell production. Unlike other fasciolids, this fluke lacks a cephalic cone or “shoulders.”
The somatic antigens of Fasciolopsis buski are crucial in triggering immune responses within the host. A prime example is the 66 kDa protein antigen, which has been demonstrated to induce the production of IgG and IgE antibodies in individuals infected with the parasite. These antigens stimulate the host’s immune system to mount robust antibody or cellular responses.
Excretory-secretory antigens constitute another essential facet of F. buski‘s interaction with its host’s immune system. These antigens can stimulate the production of antibodies or trigger cellular immune responses within the host. Mitochondrial genes are a pivotal genetic component of F. buski, encoding proteins integral to the parasite’s mitochondrial functions.
These genes encompass 12 protein-coding genes (PCGs), 22 tRNA genes, and two rRNA genes. It might be implicated in crucial processes such as energy metabolism, oxidative stress responses, and drug resistance. Also, the cytochrome c oxidase subunit-1 (cox1) gene of F. buski is a significant molecular marker employed in studies related to the parasite’s phylogenetic relationships and population genetics.
Infection with Fasciolopsis buski occurs when humans ingest contaminated water or aquatic plants containing infectious metacercariae—encysted parasite larvae. Upon ingestion, these larvae excyst within the duodenum and attach themselves to the intestinal surfaces using suckers. Over about three months, these metacercariae develop into adult worms, growing to an impressive size of 7.5 cm by 2.5 cm, making them the largest intestinal flukes known to infect humans.
These mature worms establish their residence in the proximal small bowel and produce eggs expelled through the feces. The impact of F. buski infection on the host’s health is multifaceted. The adult worms provoke harm to the intestinal mucosa through a combination of traumatic, mechanical, and toxic effects. Their presence sparks inflammation and ulceration of the mucosal lining, leading to complications such as bleeding, malabsorption, and diarrhea.
In more severe cases, the worms can instigate partial or complete bowel obstruction, resulting in intense abdominal discomfort, vomiting, nausea, and fever. Beyond the confines of the intestines, these worms may also migrate to other organs like the lungs or liver, giving rise to ectopic lesions. Remarkably, each day, the adult worms generate over 25,000 eggs.
The lifecycle of F. buski encompasses various stages, from the discharge of immature, unembryonated eggs into the intestine and stool. Over weeks, these eggs mature and hatch in water, giving rise to miracidia—tiny parasitic entities—that infiltrate a suitable snail intermediate host. Within the snail, the parasite undergoes a sequence of developmental transformations (sporocysts, rediae, and cercariae).
Subsequently, the cercariae are liberated by the snail and encyst as metacercariae on aquatic plants. When humans or other appropriate final hosts consume these plants, the metacercariae excyst within the duodenum over approximately three months and firmly attach themselves to the intestinal wall.
The consequences of F. buski infection can be wide-ranging. Bile duct blockage can occur, leading to infections or cholangitis, with fever, abdominal pain, and jaundice. Impaired nutrient absorption can result in malabsorption syndrome, accompanied by protein loss, edema, and ascites. Moreover, the worms can instigate hypersensitivity reactions in individuals, triggering responses like urticaria, angioedema, bronchospasm, or even anaphylaxis.
Fasciolopsis buski triggers a range of immune responses in the human host. The innate immune system provides initial defense. Intestinal epithelial cells form a barrier against invasion, aided by mucus that traps and obstructs parasites. Gastric acids and enzymes in the stomach can degrade ingested pathogens. Additionally, immune cells like macrophages and neutrophils perform phagocytosis of the parasite larvae, countering its establishment.
The adaptive immune response offers specific, targeted defense. T lymphocytes, including helper T cells (Th cells) and cytotoxic T cells (Tc cells), are pivotal. Th cells activate other immune cells and boost their efficacy, whereas Tc cells directly eliminate infected cells. B lymphocytes produce antibodies that neutralize the parasite, hindering its attachment and penetration. Cytokines like interferon-gamma (IFN-γ), generated by Th1 cells, enhance macrophage activity, further curbing the infection.
Fasciolopsis buski, recognized as the largest intestinal fluke afflicting humans, is responsible for causing fasciolopsiasis. The clinical expressions of fasciolopsiasis exhibit a spectrum contingent upon the intensity and duration of the infection. While some individuals may remain asymptomatic or experience only mild symptoms such as abdominal pain and diarrhea, instances of more substantial infestations can lead to greater complications.
In instances of heavy infection, these worms can precipitate severe medical concerns. Firstly, intestinal obstruction may arise, inciting intense abdominal pain, vomiting, nausea, and fever. Furthermore, the obstruction of bile ducts—critical conduits for bile transportation—can usher in complications like bile duct infections or cholangitis, characterized by symptoms such as fever, jaundice, and abdominal discomfort.
Moreover, the interference with nutrient absorption—known as malabsorption—can foster protein loss, resulting in the development of edema and ascites. Anemia may ensue due to blood loss from the intestinal wall, further exacerbating the spectrum of clinical implications. In specific scenarios, ectopic lesions might materialize, locating themselves in the intestinal wall, lungs, or other organs, extending the potential complications of this infection.
Diagnosing Fasciolopsis buski infection primarily involves identifying the parasite’s eggs or adult worms within stool samples, duodenal contents, or biliary material. The eggs of F. buski are notable for their size and oval shape, measuring between 130 to 150 μm in length and 60 to 90 μm in width. It’s important to differentiate these eggs from those of Fasciola hepatica, another liver fluke, utilizing alternative methods.
Various techniques are employed to detect F. buski eggs in stool samples:
Additionally, there are instances where the adult worms of F. buski can be located in stool samples or vomitus from infected individuals. In the diagnostic process, specific blood tests can provide valuable insights, including:
Routine blood analysis might reveal eosinophilia—an elevation in white blood cells associated with parasitic infections. Anemia—a reduction in red blood cells—and hypoalbuminemia—a decrease in a specific blood protein—might also be observable.

Both our subscription plans include Free CME/CPD AMA PRA Category 1 credits.

On course completion, you will receive a full-sized presentation quality digital certificate.
A dynamic medical simulation platform designed to train healthcare professionals and students to effectively run code situations through an immersive hands-on experience in a live, interactive 3D environment.
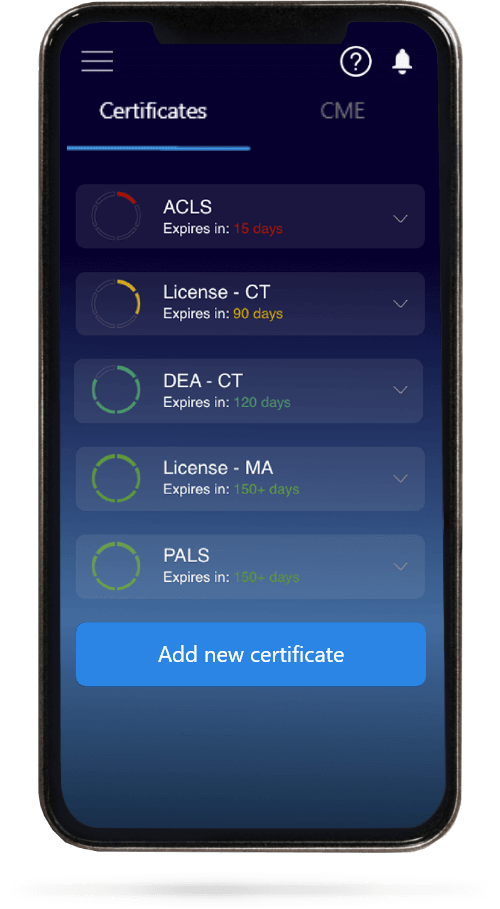
When you have your licenses, certificates and CMEs in one place, it's easier to track your career growth. You can easily share these with hospitals as well, using your medtigo app.