Pluralibacter gergoviae was first identified as a distinct species in 1980, with its name originated from the Gergovie plateau near Clermont-Ferrand University Hospital, where the type strain was isolated during a nosocomial outbreak. This bacterium is a rare human pathogen, often associated with opportunistic nosocomial infections.
It is reported to represent a small percentage (0.4%) of clinical Enterobacter isolates in some hospitals, with several risk factors contributing to infection, including prolonged healthcare stays, immunosuppression, the presence of foreign medical devices, prior antimicrobial use in patients, & extremes of age.
Beyond healthcare settings, Pluralibacter gergoviae has also been implicated in product recalls within the cosmetic industry, involving various personal care products such as eye cream, skin cream, children’s shampoo, hand cleaning paste, & cleansing wipes.
The surveillance data from the European Centre for Disease Prevention & Control (ECDC) reported 18 cases of Pluralibacter gergoviae infections in the European Union & European Economic Area (EU/EEA) in 2019, with a low notification rate of 0.004 cases per 100,000 population. Notably, most cases were imported from outside the EU/EEA, primarily originating from Asia, with higher rates observed in Luxembourg, Malta, and Cyprus.
Classification and Structure:
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Enterobacterales
Family: Enterobacteriaceae
Genus: Pluralibacter
Species: P. gergoviae
Pluralibacter gergoviae is a gram-negative bacterium with a rod-shaped morphology. Its cells typically measure 0.6 to 0.8 µm in width and 1.5 to 2.5 µm in length. Notably, it possesses a single polar flagellum that aids in its motility. This bacterium belongs to the Enterobacteriaceae family and is known for the peritrichous arrangement of fimbriae on the cell surface, which are tiny hair-like structures that assist in adhesion and colonization.
Pluralibacter gergoviae poses a significant challenge in healthcare due to its ability to build resistance to multiple antibiotics. This resistance can occur through various mechanisms, including the production of β-lactamases, efflux pumps, and mutations in target genes. Notably, Pluralibacter gergoviae can become resistant to a range of antibiotics, such as penicillins, macrolides, lincosamides, streptogramins, rifampicin, fosfomycin, fusidic acid, and cefoxitin.
This resistance complicates treatment options, potentially leading to treatment failures and increased mortality rates. Additionally, Pluralibacter gergoviae exhibits resistance to parabens, commonly used as antimicrobial agents in cosmetic products. One of the notable strains of Pluralibacter gergoviae is the type strain, P. gergoviae 16-74.
This strain was originally isolated from urine at a hospital in France and is known by various identifiers, including DSM 9245, ATCC 33028, CDC 604-77, CIP 76.01, JCM 1234, LMG 5739, and NCTC 11434. Understanding the characteristics and resistance mechanisms of Pluralibacter gergoviae strains like 16-74 is crucial for effectively managing and preventing infections caused by this opportunistic pathogen.
Pluralibacter gergoviae, as an opportunistic pathogen, poses a significant health risk to individuals, particularly those who are immunocompromised or have specific risk factors like extended hospital stays, the presence of foreign medical devices, prior antimicrobial use, or extremes of age. This bacterium can cause various human infections, including urinary tract & respiratory tract infections and bacteremia. Notably, it has been implicated in outbreaks of nosocomial bacteremias, particularly in neonatal intensive care units.
Interestingly, P. gergoviae is not confined to human infections; it has been found in diverse environmental sources such as soil, water, sewage, agricultural products like maize and grapes, and even insect guts like fruit flies and pink bollworms. One concerning aspect of this bacterium is its resistance to multiple antibiotics, which can complicate treatment efforts. It exhibits resistance to various antibiotics, including penicillins, macrolides, lincosamides, streptogramins, rifampicin, fusidic acid, fosfomycin, and cefoxitin.
Furthermore, P. gergoviae has been associated with contamination in personal care products like cosmetics, shampoos, conditioners, shower gels, and baby wipes. This bacterium resists parabens, a joint antimicrobial agent used in cosmetic products. The U.S. Food & Drug Administration (FDA) has acted in response to excessive levels of P. gergoviae found in such products, issuing warning letters to manufacturers. The FDA emphasized the potential risk of infection to sensitive groups of individuals and recommended that manufacturers implement quality controls and adhere to the FDA Good Manufacturing Practice Guidelines/Inspection Checklist for Cosmetics.
Human host defenses against Pluralibacter gergoviae involve a sophisticated interplay between innate and adaptive immunity. Innate defenses, the first line of defense, are present from birth and serve as crucial initial barriers against the bacterium. These defenses include physical barriers like the mucous membranes & skin, which act as protective shields, and chemical barriers, such as lysozyme and defensins, that exhibit antimicrobial properties.
Additionally, cellular barriers play a vital role, with phagocytes and natural killer cells at the forefront. These components of innate immunity can recognize Pluralibacter gergoviae by binding to specific antigens on the bacterium’s surface, including lipopolysaccharide (LPS), flagella, and fimbriae. Activation of various pathways, such as inflammation, complement, and cytokines, ensues, collectively working to combat the invading pathogen.
On the other hand, adaptive defenses represent the second line of defense and come into play after prior exposure to Pluralibacter gergoviae. These defenses offer specific and long-lasting protection. Humoral immunity, orchestrated by B cells, involves the production of antibodies that target the bacterium’s antigenic determinants, which can be conformational or linear epitopes. These antibodies trigger mechanisms like opsonization, neutralization, antibody-dependent cellular cytotoxicity (ADCC), and cytotoxic T lymphocyte (CTL) killing, all aimed at eliminating the bacterium. Additionally, cellular immunity, mediated by T cells, contributes to the host’s defense by recognizing and responding to the bacterium’s antigens.
In the intricate defense system, the complement system plays a critical role, comprising more than 20 proteins that become activated in a cascade-like manner upon appropriate triggering. This cascade ultimately leads to the activation of C3, a central protein, and results in the formation of the membrane attack complex.
This complex is pivotal for the bactericidal activity of serum against Gram-negative bacteria. Furthermore, complement activation yields various enzymatically cleaved proteins, with larger fragments typically binding to bacterial surfaces or components while smaller units are released. These fragments, including C3b and C3a, serve critical biological functions, such as opsonization and the recruitment of inflammatory cells.
Pluralibacter gergoviae is an infrequent human pathogen primarily associated with opportunistic nosocomial infections. The clinical manifestations of P. gergoviae infection are characterized by a range of respiratory symptoms and general signs of illness.
Patients infected with this bacterium may experience fever, chills, coughing, and difficulty breathing. These respiratory symptoms can manifest as shortness of breath, wheezing, and chest pain or discomfort. Additionally, individuals with Pluralibacter gergoviae infection may report feelings of fatigue and weakness.
Diagnosing the presence of Pluralibacter gergoviae, particularly in cosmetic products and clinical contexts, is crucial to ensuring public health and safety. Several diagnostic methods are available for this purpose:
RiboFlow Test Kits: These biomolecular test kits rapidly and efficiently detect Pluralibacter gergoviae and other Enterobacteriaceae. They work by analyzing enriched cultures or isolated colonies and can provide results within 20-25 minutes. This method is valuable for quickly identifying the presence of the bacterium in samples.
Conventional Culture Method: In this approach, a sample from the product or a patient’s bodily fluid (e.g., blood, sputum) is obtained and inoculated onto a selective agar medium. Colonies of Pluralibacter gergoviae on Tryptic Soy Agar (TSA) typically appear unpigmented, round, and smooth. This medium is often used to cultivate and isolate bacterial species, and the growth characteristics of P. gergoviae, such as its optimal growth temperature at 30°C, can aid in its identification and differentiation from other microorganisms in diagnostic procedures.
Chest X-ray Method: Chest X-rays can be employed in clinical settings, particularly when investigating pneumonia caused by Pluralibacter gergoviae in the lungs. This imaging technique helps assess the presence of pneumonia and any associated lung abnormalities caused by the bacterium. It is a valuable tool for diagnosing respiratory infections and their potential complications.
Pluralibacter gergoviae was first identified as a distinct species in 1980, with its name originated from the Gergovie plateau near Clermont-Ferrand University Hospital, where the type strain was isolated during a nosocomial outbreak. This bacterium is a rare human pathogen, often associated with opportunistic nosocomial infections.
It is reported to represent a small percentage (0.4%) of clinical Enterobacter isolates in some hospitals, with several risk factors contributing to infection, including prolonged healthcare stays, immunosuppression, the presence of foreign medical devices, prior antimicrobial use in patients, & extremes of age.
Beyond healthcare settings, Pluralibacter gergoviae has also been implicated in product recalls within the cosmetic industry, involving various personal care products such as eye cream, skin cream, children’s shampoo, hand cleaning paste, & cleansing wipes.
The surveillance data from the European Centre for Disease Prevention & Control (ECDC) reported 18 cases of Pluralibacter gergoviae infections in the European Union & European Economic Area (EU/EEA) in 2019, with a low notification rate of 0.004 cases per 100,000 population. Notably, most cases were imported from outside the EU/EEA, primarily originating from Asia, with higher rates observed in Luxembourg, Malta, and Cyprus.
Classification and Structure:
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Enterobacterales
Family: Enterobacteriaceae
Genus: Pluralibacter
Species: P. gergoviae
Pluralibacter gergoviae is a gram-negative bacterium with a rod-shaped morphology. Its cells typically measure 0.6 to 0.8 µm in width and 1.5 to 2.5 µm in length. Notably, it possesses a single polar flagellum that aids in its motility. This bacterium belongs to the Enterobacteriaceae family and is known for the peritrichous arrangement of fimbriae on the cell surface, which are tiny hair-like structures that assist in adhesion and colonization.
Pluralibacter gergoviae poses a significant challenge in healthcare due to its ability to build resistance to multiple antibiotics. This resistance can occur through various mechanisms, including the production of β-lactamases, efflux pumps, and mutations in target genes. Notably, Pluralibacter gergoviae can become resistant to a range of antibiotics, such as penicillins, macrolides, lincosamides, streptogramins, rifampicin, fosfomycin, fusidic acid, and cefoxitin.
This resistance complicates treatment options, potentially leading to treatment failures and increased mortality rates. Additionally, Pluralibacter gergoviae exhibits resistance to parabens, commonly used as antimicrobial agents in cosmetic products. One of the notable strains of Pluralibacter gergoviae is the type strain, P. gergoviae 16-74.
This strain was originally isolated from urine at a hospital in France and is known by various identifiers, including DSM 9245, ATCC 33028, CDC 604-77, CIP 76.01, JCM 1234, LMG 5739, and NCTC 11434. Understanding the characteristics and resistance mechanisms of Pluralibacter gergoviae strains like 16-74 is crucial for effectively managing and preventing infections caused by this opportunistic pathogen.
Pluralibacter gergoviae, as an opportunistic pathogen, poses a significant health risk to individuals, particularly those who are immunocompromised or have specific risk factors like extended hospital stays, the presence of foreign medical devices, prior antimicrobial use, or extremes of age. This bacterium can cause various human infections, including urinary tract & respiratory tract infections and bacteremia. Notably, it has been implicated in outbreaks of nosocomial bacteremias, particularly in neonatal intensive care units.
Interestingly, P. gergoviae is not confined to human infections; it has been found in diverse environmental sources such as soil, water, sewage, agricultural products like maize and grapes, and even insect guts like fruit flies and pink bollworms. One concerning aspect of this bacterium is its resistance to multiple antibiotics, which can complicate treatment efforts. It exhibits resistance to various antibiotics, including penicillins, macrolides, lincosamides, streptogramins, rifampicin, fusidic acid, fosfomycin, and cefoxitin.
Furthermore, P. gergoviae has been associated with contamination in personal care products like cosmetics, shampoos, conditioners, shower gels, and baby wipes. This bacterium resists parabens, a joint antimicrobial agent used in cosmetic products. The U.S. Food & Drug Administration (FDA) has acted in response to excessive levels of P. gergoviae found in such products, issuing warning letters to manufacturers. The FDA emphasized the potential risk of infection to sensitive groups of individuals and recommended that manufacturers implement quality controls and adhere to the FDA Good Manufacturing Practice Guidelines/Inspection Checklist for Cosmetics.
Human host defenses against Pluralibacter gergoviae involve a sophisticated interplay between innate and adaptive immunity. Innate defenses, the first line of defense, are present from birth and serve as crucial initial barriers against the bacterium. These defenses include physical barriers like the mucous membranes & skin, which act as protective shields, and chemical barriers, such as lysozyme and defensins, that exhibit antimicrobial properties.
Additionally, cellular barriers play a vital role, with phagocytes and natural killer cells at the forefront. These components of innate immunity can recognize Pluralibacter gergoviae by binding to specific antigens on the bacterium’s surface, including lipopolysaccharide (LPS), flagella, and fimbriae. Activation of various pathways, such as inflammation, complement, and cytokines, ensues, collectively working to combat the invading pathogen.
On the other hand, adaptive defenses represent the second line of defense and come into play after prior exposure to Pluralibacter gergoviae. These defenses offer specific and long-lasting protection. Humoral immunity, orchestrated by B cells, involves the production of antibodies that target the bacterium’s antigenic determinants, which can be conformational or linear epitopes. These antibodies trigger mechanisms like opsonization, neutralization, antibody-dependent cellular cytotoxicity (ADCC), and cytotoxic T lymphocyte (CTL) killing, all aimed at eliminating the bacterium. Additionally, cellular immunity, mediated by T cells, contributes to the host’s defense by recognizing and responding to the bacterium’s antigens.
In the intricate defense system, the complement system plays a critical role, comprising more than 20 proteins that become activated in a cascade-like manner upon appropriate triggering. This cascade ultimately leads to the activation of C3, a central protein, and results in the formation of the membrane attack complex.
This complex is pivotal for the bactericidal activity of serum against Gram-negative bacteria. Furthermore, complement activation yields various enzymatically cleaved proteins, with larger fragments typically binding to bacterial surfaces or components while smaller units are released. These fragments, including C3b and C3a, serve critical biological functions, such as opsonization and the recruitment of inflammatory cells.
Pluralibacter gergoviae is an infrequent human pathogen primarily associated with opportunistic nosocomial infections. The clinical manifestations of P. gergoviae infection are characterized by a range of respiratory symptoms and general signs of illness.
Patients infected with this bacterium may experience fever, chills, coughing, and difficulty breathing. These respiratory symptoms can manifest as shortness of breath, wheezing, and chest pain or discomfort. Additionally, individuals with Pluralibacter gergoviae infection may report feelings of fatigue and weakness.
Diagnosing the presence of Pluralibacter gergoviae, particularly in cosmetic products and clinical contexts, is crucial to ensuring public health and safety. Several diagnostic methods are available for this purpose:
RiboFlow Test Kits: These biomolecular test kits rapidly and efficiently detect Pluralibacter gergoviae and other Enterobacteriaceae. They work by analyzing enriched cultures or isolated colonies and can provide results within 20-25 minutes. This method is valuable for quickly identifying the presence of the bacterium in samples.
Conventional Culture Method: In this approach, a sample from the product or a patient’s bodily fluid (e.g., blood, sputum) is obtained and inoculated onto a selective agar medium. Colonies of Pluralibacter gergoviae on Tryptic Soy Agar (TSA) typically appear unpigmented, round, and smooth. This medium is often used to cultivate and isolate bacterial species, and the growth characteristics of P. gergoviae, such as its optimal growth temperature at 30°C, can aid in its identification and differentiation from other microorganisms in diagnostic procedures.
Chest X-ray Method: Chest X-rays can be employed in clinical settings, particularly when investigating pneumonia caused by Pluralibacter gergoviae in the lungs. This imaging technique helps assess the presence of pneumonia and any associated lung abnormalities caused by the bacterium. It is a valuable tool for diagnosing respiratory infections and their potential complications.
Pluralibacter gergoviae was first identified as a distinct species in 1980, with its name originated from the Gergovie plateau near Clermont-Ferrand University Hospital, where the type strain was isolated during a nosocomial outbreak. This bacterium is a rare human pathogen, often associated with opportunistic nosocomial infections.
It is reported to represent a small percentage (0.4%) of clinical Enterobacter isolates in some hospitals, with several risk factors contributing to infection, including prolonged healthcare stays, immunosuppression, the presence of foreign medical devices, prior antimicrobial use in patients, & extremes of age.
Beyond healthcare settings, Pluralibacter gergoviae has also been implicated in product recalls within the cosmetic industry, involving various personal care products such as eye cream, skin cream, children’s shampoo, hand cleaning paste, & cleansing wipes.
The surveillance data from the European Centre for Disease Prevention & Control (ECDC) reported 18 cases of Pluralibacter gergoviae infections in the European Union & European Economic Area (EU/EEA) in 2019, with a low notification rate of 0.004 cases per 100,000 population. Notably, most cases were imported from outside the EU/EEA, primarily originating from Asia, with higher rates observed in Luxembourg, Malta, and Cyprus.
Classification and Structure:
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Enterobacterales
Family: Enterobacteriaceae
Genus: Pluralibacter
Species: P. gergoviae
Pluralibacter gergoviae is a gram-negative bacterium with a rod-shaped morphology. Its cells typically measure 0.6 to 0.8 µm in width and 1.5 to 2.5 µm in length. Notably, it possesses a single polar flagellum that aids in its motility. This bacterium belongs to the Enterobacteriaceae family and is known for the peritrichous arrangement of fimbriae on the cell surface, which are tiny hair-like structures that assist in adhesion and colonization.
Pluralibacter gergoviae poses a significant challenge in healthcare due to its ability to build resistance to multiple antibiotics. This resistance can occur through various mechanisms, including the production of β-lactamases, efflux pumps, and mutations in target genes. Notably, Pluralibacter gergoviae can become resistant to a range of antibiotics, such as penicillins, macrolides, lincosamides, streptogramins, rifampicin, fosfomycin, fusidic acid, and cefoxitin.
This resistance complicates treatment options, potentially leading to treatment failures and increased mortality rates. Additionally, Pluralibacter gergoviae exhibits resistance to parabens, commonly used as antimicrobial agents in cosmetic products. One of the notable strains of Pluralibacter gergoviae is the type strain, P. gergoviae 16-74.
This strain was originally isolated from urine at a hospital in France and is known by various identifiers, including DSM 9245, ATCC 33028, CDC 604-77, CIP 76.01, JCM 1234, LMG 5739, and NCTC 11434. Understanding the characteristics and resistance mechanisms of Pluralibacter gergoviae strains like 16-74 is crucial for effectively managing and preventing infections caused by this opportunistic pathogen.
Pluralibacter gergoviae, as an opportunistic pathogen, poses a significant health risk to individuals, particularly those who are immunocompromised or have specific risk factors like extended hospital stays, the presence of foreign medical devices, prior antimicrobial use, or extremes of age. This bacterium can cause various human infections, including urinary tract & respiratory tract infections and bacteremia. Notably, it has been implicated in outbreaks of nosocomial bacteremias, particularly in neonatal intensive care units.
Interestingly, P. gergoviae is not confined to human infections; it has been found in diverse environmental sources such as soil, water, sewage, agricultural products like maize and grapes, and even insect guts like fruit flies and pink bollworms. One concerning aspect of this bacterium is its resistance to multiple antibiotics, which can complicate treatment efforts. It exhibits resistance to various antibiotics, including penicillins, macrolides, lincosamides, streptogramins, rifampicin, fusidic acid, fosfomycin, and cefoxitin.
Furthermore, P. gergoviae has been associated with contamination in personal care products like cosmetics, shampoos, conditioners, shower gels, and baby wipes. This bacterium resists parabens, a joint antimicrobial agent used in cosmetic products. The U.S. Food & Drug Administration (FDA) has acted in response to excessive levels of P. gergoviae found in such products, issuing warning letters to manufacturers. The FDA emphasized the potential risk of infection to sensitive groups of individuals and recommended that manufacturers implement quality controls and adhere to the FDA Good Manufacturing Practice Guidelines/Inspection Checklist for Cosmetics.
Human host defenses against Pluralibacter gergoviae involve a sophisticated interplay between innate and adaptive immunity. Innate defenses, the first line of defense, are present from birth and serve as crucial initial barriers against the bacterium. These defenses include physical barriers like the mucous membranes & skin, which act as protective shields, and chemical barriers, such as lysozyme and defensins, that exhibit antimicrobial properties.
Additionally, cellular barriers play a vital role, with phagocytes and natural killer cells at the forefront. These components of innate immunity can recognize Pluralibacter gergoviae by binding to specific antigens on the bacterium’s surface, including lipopolysaccharide (LPS), flagella, and fimbriae. Activation of various pathways, such as inflammation, complement, and cytokines, ensues, collectively working to combat the invading pathogen.
On the other hand, adaptive defenses represent the second line of defense and come into play after prior exposure to Pluralibacter gergoviae. These defenses offer specific and long-lasting protection. Humoral immunity, orchestrated by B cells, involves the production of antibodies that target the bacterium’s antigenic determinants, which can be conformational or linear epitopes. These antibodies trigger mechanisms like opsonization, neutralization, antibody-dependent cellular cytotoxicity (ADCC), and cytotoxic T lymphocyte (CTL) killing, all aimed at eliminating the bacterium. Additionally, cellular immunity, mediated by T cells, contributes to the host’s defense by recognizing and responding to the bacterium’s antigens.
In the intricate defense system, the complement system plays a critical role, comprising more than 20 proteins that become activated in a cascade-like manner upon appropriate triggering. This cascade ultimately leads to the activation of C3, a central protein, and results in the formation of the membrane attack complex.
This complex is pivotal for the bactericidal activity of serum against Gram-negative bacteria. Furthermore, complement activation yields various enzymatically cleaved proteins, with larger fragments typically binding to bacterial surfaces or components while smaller units are released. These fragments, including C3b and C3a, serve critical biological functions, such as opsonization and the recruitment of inflammatory cells.
Pluralibacter gergoviae is an infrequent human pathogen primarily associated with opportunistic nosocomial infections. The clinical manifestations of P. gergoviae infection are characterized by a range of respiratory symptoms and general signs of illness.
Patients infected with this bacterium may experience fever, chills, coughing, and difficulty breathing. These respiratory symptoms can manifest as shortness of breath, wheezing, and chest pain or discomfort. Additionally, individuals with Pluralibacter gergoviae infection may report feelings of fatigue and weakness.
Diagnosing the presence of Pluralibacter gergoviae, particularly in cosmetic products and clinical contexts, is crucial to ensuring public health and safety. Several diagnostic methods are available for this purpose:
RiboFlow Test Kits: These biomolecular test kits rapidly and efficiently detect Pluralibacter gergoviae and other Enterobacteriaceae. They work by analyzing enriched cultures or isolated colonies and can provide results within 20-25 minutes. This method is valuable for quickly identifying the presence of the bacterium in samples.
Conventional Culture Method: In this approach, a sample from the product or a patient’s bodily fluid (e.g., blood, sputum) is obtained and inoculated onto a selective agar medium. Colonies of Pluralibacter gergoviae on Tryptic Soy Agar (TSA) typically appear unpigmented, round, and smooth. This medium is often used to cultivate and isolate bacterial species, and the growth characteristics of P. gergoviae, such as its optimal growth temperature at 30°C, can aid in its identification and differentiation from other microorganisms in diagnostic procedures.
Chest X-ray Method: Chest X-rays can be employed in clinical settings, particularly when investigating pneumonia caused by Pluralibacter gergoviae in the lungs. This imaging technique helps assess the presence of pneumonia and any associated lung abnormalities caused by the bacterium. It is a valuable tool for diagnosing respiratory infections and their potential complications.

Both our subscription plans include Free CME/CPD AMA PRA Category 1 credits.

On course completion, you will receive a full-sized presentation quality digital certificate.
A dynamic medical simulation platform designed to train healthcare professionals and students to effectively run code situations through an immersive hands-on experience in a live, interactive 3D environment.
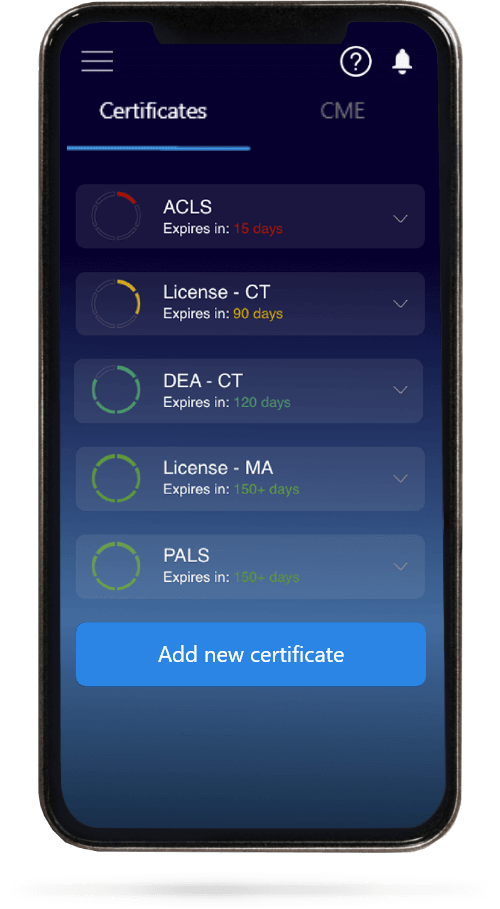
When you have your licenses, certificates and CMEs in one place, it's easier to track your career growth. You can easily share these with hospitals as well, using your medtigo app.