Puumala virus (PUUV), designated after the Finnish municipality of Puumala, where it was first identified in 1980, has a distinct epidemiology, particularly in Scandinavia & Finland. It has also been reported in other parts of Northern Europe, such as Poland and Russia. The virus is linked to the bank vole (Myodes glareolus), which acts as its reservoir. The disease developed by the Puumala virus, the nephropathia epidemic, has a cyclic pattern that coincides with the population dynamics of bank voles, recurring over three- to four-year cycles.
Inhaling dust infected with vole droppings causes humans to become infected with the Puumala virus. This mode of transmission emphasizes the significance of virus exposure in the environment. Puumala virus, unlike other ortho hantaviruses, is thought to have potentially deadly effects on its rodent host, contributing to its persistence in the vole community.
There is a high prevalence of hemorrhagic fever with renal syndrome in Europe, with over 10,000 cases identified yearly. The Puumala virus serotype causes roughly 93% of reported Hantavirus infections in Germany. Between 2002-2006, the notified incidence of HFRS ranged from 0.2 to 1.4 per 100,000 people in the province of Baden-Wuerttemberg, situated in southwestern Germany, encompassing an area of 35,742 km2 with a citizenry of 10.8 million.
By the end of March 2007, there had been a significant increase in the number of registered cases in Wuerttemberg, with 86 instances already reported, relative to an average of 12 cases during the previous five years. Puumala virus has been reported as endemic in parts of South & West Germany.
An Israeli researcher investigating the behavior of bank voles in Finland contracted the Puumala virus in August 2014, resulting in a full breakdown of her immune system. It emphasizes the severity of Puumala virus infestation in humans, especially in people who work in rodent environments.
Kingdom: Virus
Phylum: Negarnaviricota
Class: Ellioviricetes
Order: Bunyavirales
Family: Hantaviridae
Genus: Orthohantavirus
Species: Puumala virus
Puumala virus‘s viral genome is made up of three single-stranded RNA segments known as the S (small), L (large), and M (middle) and it contains viral glycoproteins that are embedded in the lipid bilayer.
PUUV is an enveloped virus with a spherical or pleomorphic shape and size of 80- 120 nm in diameter.
The most extended segment constitutes the L segment, approximately 6550 nucleotides long. It controls the RNA-dependent RNA polymerase (RdRp) necessary for viral replication.
The S segment is the smallest, measuring approximately 1830 nucleotides. It contains the genetic information for the nucleocapsid protein (N protein).
The M segment is approximately 3680 nucleotides long and encodes a glycoprotein precursor. This precursor is cleaved into two mature glycoproteins known as G1 and G2.
Puumala virus itself is classified as a serotype within the Orthohantavirus genus. Puumala virus exhibits genetic diversity and different strains or lineages. These strains are often associated with specific geographic regions or host species. Some notable strains of the Puumala virus include:
Increased capillary permeability is a critical factor in the pathogenesis of Puumala virus infections, while it primarily targets the endothelium of various organs. While the virus itself does not directly cause damage to the endothelial cells, it induces significant alterations in both the barrier function of the endothelium and the function of the infected endothelial cells.
Immune mediators contribute to the disruption of endothelial cell junctions, resulting in increased permeability of the capillaries. Consequently, fluid, proteins, and other circulating components leak from the blood vessels & accumulate in the surrounding tissues.
PUUV triggers a cascade of events that disrupt the normal physiological functions of the endothelium during infestation. These changes can lead to increased vascular permeability, compromised barrier function, and abnormal endothelial cell function. The exact mechanisms by which PUUV achieves these effects are still being investigated.
The increased capillary permeability and subsequent fluid leakage can profoundly affect the cardiovascular system, causing a drop in blood pressure and potentially leading to shock. The loss of fluid volume from the blood vessels can compromise the delivery of oxygen and nutrients to vital organs, impairing their function. Additionally, the leakage of clotting factors and platelets can contribute to forming small blood clots, further impeding blood flow.
The TNF2 allele, linked to a high TNF- producing phenotype, is linked to the HLA-B8-DR3 expanded haplotype. During PUUV infection, it may contribute to an exaggerated inflammatory response.
In PUUV infection, the broader haplotype HLA-B8-DR3 is linked to a poor prognosis. It suggests that those who carry this haplotype may be more prone to having severe symptoms and consequences. HLA-B8 and HLA-DR3 may influence T cells’ presentation of viral antigens, resulting in an exaggerated immune response or decreased virus clearance.
The HLA-B27 allele, on the other hand, has been linked to a milder phase of PUUV infection. Individuals who inherit this allele may have a more robust immune response to the virus, resulting in a milder and asymptomatic illness.
TNF-α, IL-6, and IL-10 are pro-inflammatory cytokines that trigger the immune response and facilitate viral elimination, whereas IL-10 is an anti-inflammatory cytokine that provides regulatory control for reducing immunopathology and preserving tissue homeostasis.
During PUUV infection, elevated IL-6 concentrations suggest its function in coordinating the immune response & aiding in the inflammation cascade. IL-10 concentration rises during PUUV infection, implying a counter-regulatory process to limit excessive inflammation and tissue damage. IL-10 aids in immune response modulation, preventing it from being too aggressive and harmful to the host.
Soluble TNF receptors are proteins that operate as decoy receptors, attaching to TNF- α and blocking it from interacting with cell surface receptors. Their elevated concentrations following PUUV infection suggest a regulatory mechanism to regulate TNF- overproduction and activity. Soluble TNF receptors attenuate the allergic reaction and minimize tissue damage from excess cytokine release by sequestering TNF-α.
Puumala virus primarily causes a disease known as nephropathia epidemica, which is a mild form of hemorrhagic fever with renal syndrome (HFRS). The clinical manifestations of Puumala virus infection can vary but typically include the following:
The immunofluorescence test: In this assay, the PUU Sotkamo strain of the virus, which has been grown in Vero E6 cells, is used as the antigen. The virus cells are fixed onto a microscope slide or a microtiter well. Patient serum is then added to the slide or well, and if specific antibodies against the Puumala virus are present in the serum, they will bind to the viral antigens.
To visualize this binding, fluorescein isothiocyanate (FITC)-conjugated anti-human IgG antibodies are added, which will specifically bind to the patient’s antibodies. When viewed under a fluorescence microscope, any fluorescence observed indicates the presence of PUUV-specific antibodies. For the direct enzyme immunoassays, the antigens will be diluted in phosphate-buffered saline and coated onto microtiter wells.
RT-PCR assay: RNA is extracted from the sample and converted into complementary DNA (cDNA) using reverse transcriptase enzyme. Specific primers targeting L and S segment regions like Forward Primer- 5′-GCTGCTTCCATCCATGATC-3′ and Reverse Primer- 5′-TTCCTCTTTGCCACCTTC-3′, which targets the L segment of the PUUV genome, are then used to amplify the viral genetic material through multiple cycles of PCR. The amplified DNA fragments are then analyzed, typically by gel electrophoresis or real-time PCR, to detect the presence of Puumala virus RNA.
Puumala virus (PUUV), designated after the Finnish municipality of Puumala, where it was first identified in 1980, has a distinct epidemiology, particularly in Scandinavia & Finland. It has also been reported in other parts of Northern Europe, such as Poland and Russia. The virus is linked to the bank vole (Myodes glareolus), which acts as its reservoir. The disease developed by the Puumala virus, the nephropathia epidemic, has a cyclic pattern that coincides with the population dynamics of bank voles, recurring over three- to four-year cycles.
Inhaling dust infected with vole droppings causes humans to become infected with the Puumala virus. This mode of transmission emphasizes the significance of virus exposure in the environment. Puumala virus, unlike other ortho hantaviruses, is thought to have potentially deadly effects on its rodent host, contributing to its persistence in the vole community.
There is a high prevalence of hemorrhagic fever with renal syndrome in Europe, with over 10,000 cases identified yearly. The Puumala virus serotype causes roughly 93% of reported Hantavirus infections in Germany. Between 2002-2006, the notified incidence of HFRS ranged from 0.2 to 1.4 per 100,000 people in the province of Baden-Wuerttemberg, situated in southwestern Germany, encompassing an area of 35,742 km2 with a citizenry of 10.8 million.
By the end of March 2007, there had been a significant increase in the number of registered cases in Wuerttemberg, with 86 instances already reported, relative to an average of 12 cases during the previous five years. Puumala virus has been reported as endemic in parts of South & West Germany.
An Israeli researcher investigating the behavior of bank voles in Finland contracted the Puumala virus in August 2014, resulting in a full breakdown of her immune system. It emphasizes the severity of Puumala virus infestation in humans, especially in people who work in rodent environments.
Kingdom: Virus
Phylum: Negarnaviricota
Class: Ellioviricetes
Order: Bunyavirales
Family: Hantaviridae
Genus: Orthohantavirus
Species: Puumala virus
Puumala virus‘s viral genome is made up of three single-stranded RNA segments known as the S (small), L (large), and M (middle) and it contains viral glycoproteins that are embedded in the lipid bilayer.
PUUV is an enveloped virus with a spherical or pleomorphic shape and size of 80- 120 nm in diameter.
The most extended segment constitutes the L segment, approximately 6550 nucleotides long. It controls the RNA-dependent RNA polymerase (RdRp) necessary for viral replication.
The S segment is the smallest, measuring approximately 1830 nucleotides. It contains the genetic information for the nucleocapsid protein (N protein).
The M segment is approximately 3680 nucleotides long and encodes a glycoprotein precursor. This precursor is cleaved into two mature glycoproteins known as G1 and G2.
Puumala virus itself is classified as a serotype within the Orthohantavirus genus. Puumala virus exhibits genetic diversity and different strains or lineages. These strains are often associated with specific geographic regions or host species. Some notable strains of the Puumala virus include:
Increased capillary permeability is a critical factor in the pathogenesis of Puumala virus infections, while it primarily targets the endothelium of various organs. While the virus itself does not directly cause damage to the endothelial cells, it induces significant alterations in both the barrier function of the endothelium and the function of the infected endothelial cells.
Immune mediators contribute to the disruption of endothelial cell junctions, resulting in increased permeability of the capillaries. Consequently, fluid, proteins, and other circulating components leak from the blood vessels & accumulate in the surrounding tissues.
PUUV triggers a cascade of events that disrupt the normal physiological functions of the endothelium during infestation. These changes can lead to increased vascular permeability, compromised barrier function, and abnormal endothelial cell function. The exact mechanisms by which PUUV achieves these effects are still being investigated.
The increased capillary permeability and subsequent fluid leakage can profoundly affect the cardiovascular system, causing a drop in blood pressure and potentially leading to shock. The loss of fluid volume from the blood vessels can compromise the delivery of oxygen and nutrients to vital organs, impairing their function. Additionally, the leakage of clotting factors and platelets can contribute to forming small blood clots, further impeding blood flow.
The TNF2 allele, linked to a high TNF- producing phenotype, is linked to the HLA-B8-DR3 expanded haplotype. During PUUV infection, it may contribute to an exaggerated inflammatory response.
In PUUV infection, the broader haplotype HLA-B8-DR3 is linked to a poor prognosis. It suggests that those who carry this haplotype may be more prone to having severe symptoms and consequences. HLA-B8 and HLA-DR3 may influence T cells’ presentation of viral antigens, resulting in an exaggerated immune response or decreased virus clearance.
The HLA-B27 allele, on the other hand, has been linked to a milder phase of PUUV infection. Individuals who inherit this allele may have a more robust immune response to the virus, resulting in a milder and asymptomatic illness.
TNF-α, IL-6, and IL-10 are pro-inflammatory cytokines that trigger the immune response and facilitate viral elimination, whereas IL-10 is an anti-inflammatory cytokine that provides regulatory control for reducing immunopathology and preserving tissue homeostasis.
During PUUV infection, elevated IL-6 concentrations suggest its function in coordinating the immune response & aiding in the inflammation cascade. IL-10 concentration rises during PUUV infection, implying a counter-regulatory process to limit excessive inflammation and tissue damage. IL-10 aids in immune response modulation, preventing it from being too aggressive and harmful to the host.
Soluble TNF receptors are proteins that operate as decoy receptors, attaching to TNF- α and blocking it from interacting with cell surface receptors. Their elevated concentrations following PUUV infection suggest a regulatory mechanism to regulate TNF- overproduction and activity. Soluble TNF receptors attenuate the allergic reaction and minimize tissue damage from excess cytokine release by sequestering TNF-α.
Puumala virus primarily causes a disease known as nephropathia epidemica, which is a mild form of hemorrhagic fever with renal syndrome (HFRS). The clinical manifestations of Puumala virus infection can vary but typically include the following:
The immunofluorescence test: In this assay, the PUU Sotkamo strain of the virus, which has been grown in Vero E6 cells, is used as the antigen. The virus cells are fixed onto a microscope slide or a microtiter well. Patient serum is then added to the slide or well, and if specific antibodies against the Puumala virus are present in the serum, they will bind to the viral antigens.
To visualize this binding, fluorescein isothiocyanate (FITC)-conjugated anti-human IgG antibodies are added, which will specifically bind to the patient’s antibodies. When viewed under a fluorescence microscope, any fluorescence observed indicates the presence of PUUV-specific antibodies. For the direct enzyme immunoassays, the antigens will be diluted in phosphate-buffered saline and coated onto microtiter wells.
RT-PCR assay: RNA is extracted from the sample and converted into complementary DNA (cDNA) using reverse transcriptase enzyme. Specific primers targeting L and S segment regions like Forward Primer- 5′-GCTGCTTCCATCCATGATC-3′ and Reverse Primer- 5′-TTCCTCTTTGCCACCTTC-3′, which targets the L segment of the PUUV genome, are then used to amplify the viral genetic material through multiple cycles of PCR. The amplified DNA fragments are then analyzed, typically by gel electrophoresis or real-time PCR, to detect the presence of Puumala virus RNA.
Puumala virus (PUUV), designated after the Finnish municipality of Puumala, where it was first identified in 1980, has a distinct epidemiology, particularly in Scandinavia & Finland. It has also been reported in other parts of Northern Europe, such as Poland and Russia. The virus is linked to the bank vole (Myodes glareolus), which acts as its reservoir. The disease developed by the Puumala virus, the nephropathia epidemic, has a cyclic pattern that coincides with the population dynamics of bank voles, recurring over three- to four-year cycles.
Inhaling dust infected with vole droppings causes humans to become infected with the Puumala virus. This mode of transmission emphasizes the significance of virus exposure in the environment. Puumala virus, unlike other ortho hantaviruses, is thought to have potentially deadly effects on its rodent host, contributing to its persistence in the vole community.
There is a high prevalence of hemorrhagic fever with renal syndrome in Europe, with over 10,000 cases identified yearly. The Puumala virus serotype causes roughly 93% of reported Hantavirus infections in Germany. Between 2002-2006, the notified incidence of HFRS ranged from 0.2 to 1.4 per 100,000 people in the province of Baden-Wuerttemberg, situated in southwestern Germany, encompassing an area of 35,742 km2 with a citizenry of 10.8 million.
By the end of March 2007, there had been a significant increase in the number of registered cases in Wuerttemberg, with 86 instances already reported, relative to an average of 12 cases during the previous five years. Puumala virus has been reported as endemic in parts of South & West Germany.
An Israeli researcher investigating the behavior of bank voles in Finland contracted the Puumala virus in August 2014, resulting in a full breakdown of her immune system. It emphasizes the severity of Puumala virus infestation in humans, especially in people who work in rodent environments.
Kingdom: Virus
Phylum: Negarnaviricota
Class: Ellioviricetes
Order: Bunyavirales
Family: Hantaviridae
Genus: Orthohantavirus
Species: Puumala virus
Puumala virus‘s viral genome is made up of three single-stranded RNA segments known as the S (small), L (large), and M (middle) and it contains viral glycoproteins that are embedded in the lipid bilayer.
PUUV is an enveloped virus with a spherical or pleomorphic shape and size of 80- 120 nm in diameter.
The most extended segment constitutes the L segment, approximately 6550 nucleotides long. It controls the RNA-dependent RNA polymerase (RdRp) necessary for viral replication.
The S segment is the smallest, measuring approximately 1830 nucleotides. It contains the genetic information for the nucleocapsid protein (N protein).
The M segment is approximately 3680 nucleotides long and encodes a glycoprotein precursor. This precursor is cleaved into two mature glycoproteins known as G1 and G2.
Puumala virus itself is classified as a serotype within the Orthohantavirus genus. Puumala virus exhibits genetic diversity and different strains or lineages. These strains are often associated with specific geographic regions or host species. Some notable strains of the Puumala virus include:
Increased capillary permeability is a critical factor in the pathogenesis of Puumala virus infections, while it primarily targets the endothelium of various organs. While the virus itself does not directly cause damage to the endothelial cells, it induces significant alterations in both the barrier function of the endothelium and the function of the infected endothelial cells.
Immune mediators contribute to the disruption of endothelial cell junctions, resulting in increased permeability of the capillaries. Consequently, fluid, proteins, and other circulating components leak from the blood vessels & accumulate in the surrounding tissues.
PUUV triggers a cascade of events that disrupt the normal physiological functions of the endothelium during infestation. These changes can lead to increased vascular permeability, compromised barrier function, and abnormal endothelial cell function. The exact mechanisms by which PUUV achieves these effects are still being investigated.
The increased capillary permeability and subsequent fluid leakage can profoundly affect the cardiovascular system, causing a drop in blood pressure and potentially leading to shock. The loss of fluid volume from the blood vessels can compromise the delivery of oxygen and nutrients to vital organs, impairing their function. Additionally, the leakage of clotting factors and platelets can contribute to forming small blood clots, further impeding blood flow.
The TNF2 allele, linked to a high TNF- producing phenotype, is linked to the HLA-B8-DR3 expanded haplotype. During PUUV infection, it may contribute to an exaggerated inflammatory response.
In PUUV infection, the broader haplotype HLA-B8-DR3 is linked to a poor prognosis. It suggests that those who carry this haplotype may be more prone to having severe symptoms and consequences. HLA-B8 and HLA-DR3 may influence T cells’ presentation of viral antigens, resulting in an exaggerated immune response or decreased virus clearance.
The HLA-B27 allele, on the other hand, has been linked to a milder phase of PUUV infection. Individuals who inherit this allele may have a more robust immune response to the virus, resulting in a milder and asymptomatic illness.
TNF-α, IL-6, and IL-10 are pro-inflammatory cytokines that trigger the immune response and facilitate viral elimination, whereas IL-10 is an anti-inflammatory cytokine that provides regulatory control for reducing immunopathology and preserving tissue homeostasis.
During PUUV infection, elevated IL-6 concentrations suggest its function in coordinating the immune response & aiding in the inflammation cascade. IL-10 concentration rises during PUUV infection, implying a counter-regulatory process to limit excessive inflammation and tissue damage. IL-10 aids in immune response modulation, preventing it from being too aggressive and harmful to the host.
Soluble TNF receptors are proteins that operate as decoy receptors, attaching to TNF- α and blocking it from interacting with cell surface receptors. Their elevated concentrations following PUUV infection suggest a regulatory mechanism to regulate TNF- overproduction and activity. Soluble TNF receptors attenuate the allergic reaction and minimize tissue damage from excess cytokine release by sequestering TNF-α.
Puumala virus primarily causes a disease known as nephropathia epidemica, which is a mild form of hemorrhagic fever with renal syndrome (HFRS). The clinical manifestations of Puumala virus infection can vary but typically include the following:
The immunofluorescence test: In this assay, the PUU Sotkamo strain of the virus, which has been grown in Vero E6 cells, is used as the antigen. The virus cells are fixed onto a microscope slide or a microtiter well. Patient serum is then added to the slide or well, and if specific antibodies against the Puumala virus are present in the serum, they will bind to the viral antigens.
To visualize this binding, fluorescein isothiocyanate (FITC)-conjugated anti-human IgG antibodies are added, which will specifically bind to the patient’s antibodies. When viewed under a fluorescence microscope, any fluorescence observed indicates the presence of PUUV-specific antibodies. For the direct enzyme immunoassays, the antigens will be diluted in phosphate-buffered saline and coated onto microtiter wells.
RT-PCR assay: RNA is extracted from the sample and converted into complementary DNA (cDNA) using reverse transcriptase enzyme. Specific primers targeting L and S segment regions like Forward Primer- 5′-GCTGCTTCCATCCATGATC-3′ and Reverse Primer- 5′-TTCCTCTTTGCCACCTTC-3′, which targets the L segment of the PUUV genome, are then used to amplify the viral genetic material through multiple cycles of PCR. The amplified DNA fragments are then analyzed, typically by gel electrophoresis or real-time PCR, to detect the presence of Puumala virus RNA.

Both our subscription plans include Free CME/CPD AMA PRA Category 1 credits.

On course completion, you will receive a full-sized presentation quality digital certificate.
A dynamic medical simulation platform designed to train healthcare professionals and students to effectively run code situations through an immersive hands-on experience in a live, interactive 3D environment.
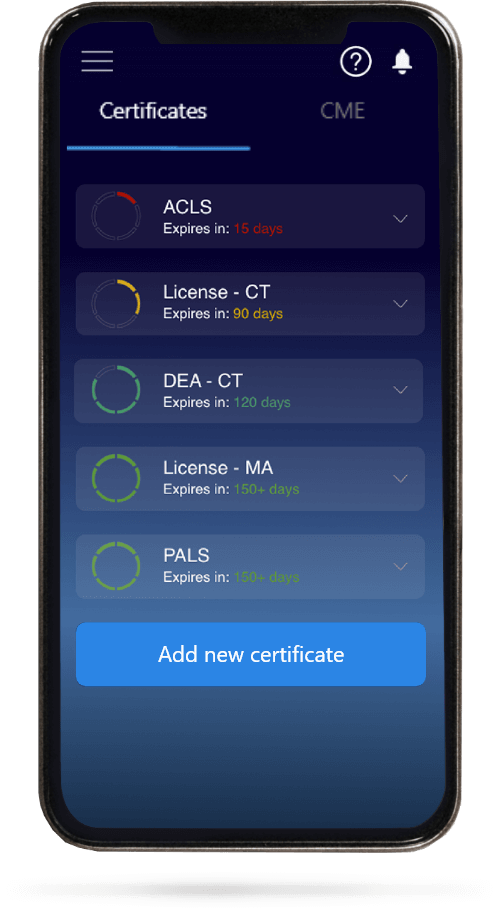
When you have your licenses, certificates and CMEs in one place, it's easier to track your career growth. You can easily share these with hospitals as well, using your medtigo app.