The worldwide cases of Providencia stuartii infections are not precisely known due to limited data, but some estimates can be derived from various studies. For instance, a study from Taiwan reported 24 cases of P. stuartii bacteremia over 10 years, indicating its presence in that region.
Similarly, a study from Spain documented 19 cases of Providencia stuartii urinary tract infections over 5 years, highlighting its occurrence in different parts of the world. Moreover, a significant outbreak of carbapenem-resistant P. stuartii infections affected 27 patients in an intensive care unit in Brazil, indicating the potential for larger-scale outbreaks in healthcare settings.
Providencia stuartii infections have been globally reported, with a higher prevalence in developed countries. The most common type of infection is urinary tract infection, followed by wound infections. Notably, P. stuartii infections pose a challenge for treatment, as the bacterium often exhibits resistance to multiple antibiotics, including beta-lactams, fluoroquinolones, aminoglycosides, sulfonamides, and trimethoprim. This antibiotic resistance significantly hampers therapeutic options, leading to prolonged and complicated treatment regimens.
The mortality rate of Providencia stuartii infections varies based on the type and severity of the infection, ranging from 10% to 45%. This wide range underscores the importance of timely and appropriate management to improve patient outcomes. The primary sources of infection are the patient’s flora or the environment. While person-to-person transmission is possible, it is relatively uncommon.
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Enterobacterales
Family: Morganellaceae
Genus: Providencia
Species: Providencia stuartii
Providencia stuartii is a straight rod-shaped bacterium with a typical size of approximately 0.6-0.8 μm in diameter and 1.5-2.5 μm in length. The cell envelope of Providencia stuartii consists of three layers: a thin peptidoglycan cell wall, a cytoplasmic membrane, and an outer membrane. The cytoplasmic membrane is a phospholipid bilayer that surrounds the cytoplasm and acts as a selective barrier.
LPS (lipopoly saccharide) molecules are present in the outer leaflet of the outer membrane. They contain lipid A, a core oligosaccharide, and an O antigen. Providencia stuartii is motile due to the presence of flagella. These whip-like appendages enable the bacterium to move in liquid environments.
ESBLs: Extended-spectrum beta-lactamases (ESBLs) are enzymes produced by Providencia stuartii that confer resistance to beta-lactam antibiotics, including penicillins, cephalosporins, and monobactams. These ESBLs are often encoded by plasmids, allowing for the horizontal transfer of antibiotic-resistance genes among bacteria.
Integrons: Providencia stuartii can harbor integrons, which are genetic elements capable of capturing and expressing antibiotic resistance genes from various sources. Integrons play a significant role in conferring resistance to multiple classes of antibiotics, such as aminoglycosides, fluoroquinolones, sulfonamides, and trimethoprim.
Strains of Providencia stuartii:
P. stuartii MRSN 2154: This strain of Providencia stuartii was isolated from a wound infection in the United States. With a genome size of 4.7 Mb and containing 4,318 protein-coding genes, it carries multiple antibiotic resistance genes, such as blaTEM-1, blaSHV-12, blaOXA-9, blaNDM-1, and aac(6′)-Ib-cr.
P. stuartii RYC492: Isolated from human urine in Spain, this strain has a genome size of 4.6 Mb and contains 4,232 protein-coding genes.
P. stuartii ATCC 25827: Isolated from human feces in the United States, this strain has a genome size of 4.5 Mb and contains 4,122 protein-coding genes.
P. stuartii DSM 3043: Isolated from human urine in Germany, this strain has a genome size of 4.5 Mb and contains 4,137 protein-coding genes.
Providencia stuartii employs several fundamental mechanisms to initiate and establish infections in the host. Adhesion and colonization are facilitated by specific outer membrane proteins like fimbriae, pili, and lipopolysaccharides, allowing the bacterium to adhere to various host tissues, such as those in the urinary tract, respiratory tract, skin, or wounds.
Once adhered, Providencia stuartii can invade host epithelial cells and escape from phagolysosomes using enzymes like phospholipase A, hemolysin, and protease. These enzymes damage host membranes, leading to cell lysis, inflammation, and necrosis.
Motility, enabled by peritrichous flagella, allows Providencia stuartii to swarm on semi-solid surfaces and efficiently colonize host tissues. Additionally, the bacterium forms biofilms that protect host immune responses and antimicrobial agents, leading to persistent infections.
Antibiotic resistance is a significant concern with Providencia stuartii, as it can produce extended-spectrum beta-lactamases (ESBLs) that hydrolyze beta-lactam antibiotics, rendering them ineffective. Furthermore, Providencia stuartii can harbor integrons that capture and express antibiotic-resistance genes, leading to resistance against multiple classes of antibiotics.
Another tactic Providencia stuartii uses involves the modulation of porins, which affects the outer membrane’s permeability and can influence the bacterium’s susceptibility to antibiotics. In severe cases or immunocompromised individuals, Providencia stuartii can disseminate from the initial infection site to other body parts through the bloodstream, leading to systemic infections like pneumonia or meningitis.
In the battle against Providencia stuartii, the innate immune system deploys an array of robust mechanisms to halt the infection at its early stages. Macrophages and epithelial cells produce nitric oxide (NO). This potent molecule can wreak havoc on bacterial membranes and DNA, effectively hindering the growth of P. stuartii both in laboratory settings and within the host’s body.
Additionally, epithelial cells and neutrophils secrete antimicrobial peptides, such as defensins, cathelicidins, and LL-37. These act as small yet formidable proteins that disrupt bacterial membranes and interfere with their essential metabolic processes. Furthermore, the complement system, a group of serum proteins, gets activated to initiate a cascade of reactions.
This process culminates in forming membrane attack complexes (MACs) on the bacterial surface. MACs bore holes into the bacterial membrane, inducing cell lysis and destruction. Additionally, the complement system enhances the process of phagocytosis, whereby immune cells engulf and dispose of the bacteria, further fortifying the body’s defenses.
As the infection progresses, the adaptive immune system joins the fray, arming itself with specialized tactics to counter P. stuartii. B lymphocytes are deployed to produce antibodies that recognize and bind to specific antigens present on the surface of the bacteria. These antibodies serve many purposes – they neutralize the bacteria, facilitate phagocytosis, and enhance the opsonization process by interacting with Fc receptors on phagocytes.
Simultaneously, antigen-presenting cells activate T lymphocytes, divided into two primary subsets: helper T cells (Th) and cytotoxic T cells (Tc). Helper T cells orchestrate the immune response by releasing cytokines that regulate the activities of various immune cells, like B cells, neutrophils, & macrophages. This coordination is crucial in combating the infection effectively.
Providencia stuartii is a significant pathogen known to cause various infections, with urinary tract infections being one of the most common in catheterized patients, particularly in hospitals and long-term care facilities. However, this opportunistic pathogen can also lead to other severe infections, including wound infections, pneumonia, bacteremia, meningitis, and Fournier’s gangrene, particularly in critically ill patients or those with underlying medical conditions. Clinical manifestations of Providencia stuartii infections often include fever, tachycardia, hypotension, suprapubic tenderness, and flank pain.
In elderly individuals, who are more prone to having long-term indwelling urinary catheters, the risk of P. stuartii infections is high. The bacterium has also been found to cause neonatal diarrhea in dairy cows. In humans, Providencia stuartii can be isolated from various sources, including urine (most common), stool, blood, sputum, skin, and wound cultures. In cases of P. stuartii sepsis, the primary origin is often urinary. Notably, it is the most common cause of purple urine bag syndrome.
Physical examination of patients with Providencia stuartii bloodstream infection typically reveals symptoms like fever, rapid heart rate (tachycardia), and low blood pressure (hypotension). Due to its opportunistic nature and association with invasive medical procedures, healthcare providers must be vigilant in diagnosing and managing Providencia stuartii infections, especially in vulnerable populations.
Identifying Providencia stuartii from other bacteria, particularly within the Enterobacteriaceae family, involves a series of diagnostic tests that showcase its distinct characteristics:
Culture test: Providencia stuartii displays non-lactose fermenting properties, forming colorless colonies on MacConkey agar due to lactose fermentation’s lack of acid production.
Gram stain: Providencia stuartii is recognized as Gram-negative bacilli, exhibiting a pink color after counterstaining with safranin rather than retaining the purple dye.
Motility test: Providencia stuartii‘s motility is evident, as it possesses flagella that allow it to move in liquid media or semi-solid agar, leading to diffuse growth or turbidity in motility test medium or SIM agar.
Voges-Proskauer (VP) test: Providencia stuartii exhibits a negative VP test, as it does not produce acetoin or 2,3-butanediol from glucose fermentation, resulting in the absence of a red complex when Barritt’s reagent is applied.
Oxidase test: Providencia stuartii is characterized as oxidase negative, indicating the absence of the enzyme cytochrome c oxidase.
Indole test: Providencia stuartii exhibits a positive indole test, demonstrating its capability to produce indole from the amino acid tryptophan, resulting in a distinct red ring with Kovac’s reagent.
Methyl red (MR) test: Providencia stuartii yields a positive MR test, indicating its ability to produce mixed acids from glucose fermentation, which lowers the medium’s pH below 4.4, causing a color shift from yellow to red with methyl red.
Urease test: Providencia stuartii tests negative for urease production, indicating the absence of hydrolysis of urea to ammonia and carbon dioxide, thus preventing a color shift from yellow to pink on urea agar or broth.
The worldwide cases of Providencia stuartii infections are not precisely known due to limited data, but some estimates can be derived from various studies. For instance, a study from Taiwan reported 24 cases of P. stuartii bacteremia over 10 years, indicating its presence in that region.
Similarly, a study from Spain documented 19 cases of Providencia stuartii urinary tract infections over 5 years, highlighting its occurrence in different parts of the world. Moreover, a significant outbreak of carbapenem-resistant P. stuartii infections affected 27 patients in an intensive care unit in Brazil, indicating the potential for larger-scale outbreaks in healthcare settings.
Providencia stuartii infections have been globally reported, with a higher prevalence in developed countries. The most common type of infection is urinary tract infection, followed by wound infections. Notably, P. stuartii infections pose a challenge for treatment, as the bacterium often exhibits resistance to multiple antibiotics, including beta-lactams, fluoroquinolones, aminoglycosides, sulfonamides, and trimethoprim. This antibiotic resistance significantly hampers therapeutic options, leading to prolonged and complicated treatment regimens.
The mortality rate of Providencia stuartii infections varies based on the type and severity of the infection, ranging from 10% to 45%. This wide range underscores the importance of timely and appropriate management to improve patient outcomes. The primary sources of infection are the patient’s flora or the environment. While person-to-person transmission is possible, it is relatively uncommon.
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Enterobacterales
Family: Morganellaceae
Genus: Providencia
Species: Providencia stuartii
Providencia stuartii is a straight rod-shaped bacterium with a typical size of approximately 0.6-0.8 μm in diameter and 1.5-2.5 μm in length. The cell envelope of Providencia stuartii consists of three layers: a thin peptidoglycan cell wall, a cytoplasmic membrane, and an outer membrane. The cytoplasmic membrane is a phospholipid bilayer that surrounds the cytoplasm and acts as a selective barrier.
LPS (lipopoly saccharide) molecules are present in the outer leaflet of the outer membrane. They contain lipid A, a core oligosaccharide, and an O antigen. Providencia stuartii is motile due to the presence of flagella. These whip-like appendages enable the bacterium to move in liquid environments.
ESBLs: Extended-spectrum beta-lactamases (ESBLs) are enzymes produced by Providencia stuartii that confer resistance to beta-lactam antibiotics, including penicillins, cephalosporins, and monobactams. These ESBLs are often encoded by plasmids, allowing for the horizontal transfer of antibiotic-resistance genes among bacteria.
Integrons: Providencia stuartii can harbor integrons, which are genetic elements capable of capturing and expressing antibiotic resistance genes from various sources. Integrons play a significant role in conferring resistance to multiple classes of antibiotics, such as aminoglycosides, fluoroquinolones, sulfonamides, and trimethoprim.
Strains of Providencia stuartii:
P. stuartii MRSN 2154: This strain of Providencia stuartii was isolated from a wound infection in the United States. With a genome size of 4.7 Mb and containing 4,318 protein-coding genes, it carries multiple antibiotic resistance genes, such as blaTEM-1, blaSHV-12, blaOXA-9, blaNDM-1, and aac(6′)-Ib-cr.
P. stuartii RYC492: Isolated from human urine in Spain, this strain has a genome size of 4.6 Mb and contains 4,232 protein-coding genes.
P. stuartii ATCC 25827: Isolated from human feces in the United States, this strain has a genome size of 4.5 Mb and contains 4,122 protein-coding genes.
P. stuartii DSM 3043: Isolated from human urine in Germany, this strain has a genome size of 4.5 Mb and contains 4,137 protein-coding genes.
Providencia stuartii employs several fundamental mechanisms to initiate and establish infections in the host. Adhesion and colonization are facilitated by specific outer membrane proteins like fimbriae, pili, and lipopolysaccharides, allowing the bacterium to adhere to various host tissues, such as those in the urinary tract, respiratory tract, skin, or wounds.
Once adhered, Providencia stuartii can invade host epithelial cells and escape from phagolysosomes using enzymes like phospholipase A, hemolysin, and protease. These enzymes damage host membranes, leading to cell lysis, inflammation, and necrosis.
Motility, enabled by peritrichous flagella, allows Providencia stuartii to swarm on semi-solid surfaces and efficiently colonize host tissues. Additionally, the bacterium forms biofilms that protect host immune responses and antimicrobial agents, leading to persistent infections.
Antibiotic resistance is a significant concern with Providencia stuartii, as it can produce extended-spectrum beta-lactamases (ESBLs) that hydrolyze beta-lactam antibiotics, rendering them ineffective. Furthermore, Providencia stuartii can harbor integrons that capture and express antibiotic-resistance genes, leading to resistance against multiple classes of antibiotics.
Another tactic Providencia stuartii uses involves the modulation of porins, which affects the outer membrane’s permeability and can influence the bacterium’s susceptibility to antibiotics. In severe cases or immunocompromised individuals, Providencia stuartii can disseminate from the initial infection site to other body parts through the bloodstream, leading to systemic infections like pneumonia or meningitis.
In the battle against Providencia stuartii, the innate immune system deploys an array of robust mechanisms to halt the infection at its early stages. Macrophages and epithelial cells produce nitric oxide (NO). This potent molecule can wreak havoc on bacterial membranes and DNA, effectively hindering the growth of P. stuartii both in laboratory settings and within the host’s body.
Additionally, epithelial cells and neutrophils secrete antimicrobial peptides, such as defensins, cathelicidins, and LL-37. These act as small yet formidable proteins that disrupt bacterial membranes and interfere with their essential metabolic processes. Furthermore, the complement system, a group of serum proteins, gets activated to initiate a cascade of reactions.
This process culminates in forming membrane attack complexes (MACs) on the bacterial surface. MACs bore holes into the bacterial membrane, inducing cell lysis and destruction. Additionally, the complement system enhances the process of phagocytosis, whereby immune cells engulf and dispose of the bacteria, further fortifying the body’s defenses.
As the infection progresses, the adaptive immune system joins the fray, arming itself with specialized tactics to counter P. stuartii. B lymphocytes are deployed to produce antibodies that recognize and bind to specific antigens present on the surface of the bacteria. These antibodies serve many purposes – they neutralize the bacteria, facilitate phagocytosis, and enhance the opsonization process by interacting with Fc receptors on phagocytes.
Simultaneously, antigen-presenting cells activate T lymphocytes, divided into two primary subsets: helper T cells (Th) and cytotoxic T cells (Tc). Helper T cells orchestrate the immune response by releasing cytokines that regulate the activities of various immune cells, like B cells, neutrophils, & macrophages. This coordination is crucial in combating the infection effectively.
Providencia stuartii is a significant pathogen known to cause various infections, with urinary tract infections being one of the most common in catheterized patients, particularly in hospitals and long-term care facilities. However, this opportunistic pathogen can also lead to other severe infections, including wound infections, pneumonia, bacteremia, meningitis, and Fournier’s gangrene, particularly in critically ill patients or those with underlying medical conditions. Clinical manifestations of Providencia stuartii infections often include fever, tachycardia, hypotension, suprapubic tenderness, and flank pain.
In elderly individuals, who are more prone to having long-term indwelling urinary catheters, the risk of P. stuartii infections is high. The bacterium has also been found to cause neonatal diarrhea in dairy cows. In humans, Providencia stuartii can be isolated from various sources, including urine (most common), stool, blood, sputum, skin, and wound cultures. In cases of P. stuartii sepsis, the primary origin is often urinary. Notably, it is the most common cause of purple urine bag syndrome.
Physical examination of patients with Providencia stuartii bloodstream infection typically reveals symptoms like fever, rapid heart rate (tachycardia), and low blood pressure (hypotension). Due to its opportunistic nature and association with invasive medical procedures, healthcare providers must be vigilant in diagnosing and managing Providencia stuartii infections, especially in vulnerable populations.
Identifying Providencia stuartii from other bacteria, particularly within the Enterobacteriaceae family, involves a series of diagnostic tests that showcase its distinct characteristics:
Culture test: Providencia stuartii displays non-lactose fermenting properties, forming colorless colonies on MacConkey agar due to lactose fermentation’s lack of acid production.
Gram stain: Providencia stuartii is recognized as Gram-negative bacilli, exhibiting a pink color after counterstaining with safranin rather than retaining the purple dye.
Motility test: Providencia stuartii‘s motility is evident, as it possesses flagella that allow it to move in liquid media or semi-solid agar, leading to diffuse growth or turbidity in motility test medium or SIM agar.
Voges-Proskauer (VP) test: Providencia stuartii exhibits a negative VP test, as it does not produce acetoin or 2,3-butanediol from glucose fermentation, resulting in the absence of a red complex when Barritt’s reagent is applied.
Oxidase test: Providencia stuartii is characterized as oxidase negative, indicating the absence of the enzyme cytochrome c oxidase.
Indole test: Providencia stuartii exhibits a positive indole test, demonstrating its capability to produce indole from the amino acid tryptophan, resulting in a distinct red ring with Kovac’s reagent.
Methyl red (MR) test: Providencia stuartii yields a positive MR test, indicating its ability to produce mixed acids from glucose fermentation, which lowers the medium’s pH below 4.4, causing a color shift from yellow to red with methyl red.
Urease test: Providencia stuartii tests negative for urease production, indicating the absence of hydrolysis of urea to ammonia and carbon dioxide, thus preventing a color shift from yellow to pink on urea agar or broth.
The worldwide cases of Providencia stuartii infections are not precisely known due to limited data, but some estimates can be derived from various studies. For instance, a study from Taiwan reported 24 cases of P. stuartii bacteremia over 10 years, indicating its presence in that region.
Similarly, a study from Spain documented 19 cases of Providencia stuartii urinary tract infections over 5 years, highlighting its occurrence in different parts of the world. Moreover, a significant outbreak of carbapenem-resistant P. stuartii infections affected 27 patients in an intensive care unit in Brazil, indicating the potential for larger-scale outbreaks in healthcare settings.
Providencia stuartii infections have been globally reported, with a higher prevalence in developed countries. The most common type of infection is urinary tract infection, followed by wound infections. Notably, P. stuartii infections pose a challenge for treatment, as the bacterium often exhibits resistance to multiple antibiotics, including beta-lactams, fluoroquinolones, aminoglycosides, sulfonamides, and trimethoprim. This antibiotic resistance significantly hampers therapeutic options, leading to prolonged and complicated treatment regimens.
The mortality rate of Providencia stuartii infections varies based on the type and severity of the infection, ranging from 10% to 45%. This wide range underscores the importance of timely and appropriate management to improve patient outcomes. The primary sources of infection are the patient’s flora or the environment. While person-to-person transmission is possible, it is relatively uncommon.
Kingdom: Bacteria
Phylum: Pseudomonadota
Class: Gammaproteobacteria
Order: Enterobacterales
Family: Morganellaceae
Genus: Providencia
Species: Providencia stuartii
Providencia stuartii is a straight rod-shaped bacterium with a typical size of approximately 0.6-0.8 μm in diameter and 1.5-2.5 μm in length. The cell envelope of Providencia stuartii consists of three layers: a thin peptidoglycan cell wall, a cytoplasmic membrane, and an outer membrane. The cytoplasmic membrane is a phospholipid bilayer that surrounds the cytoplasm and acts as a selective barrier.
LPS (lipopoly saccharide) molecules are present in the outer leaflet of the outer membrane. They contain lipid A, a core oligosaccharide, and an O antigen. Providencia stuartii is motile due to the presence of flagella. These whip-like appendages enable the bacterium to move in liquid environments.
ESBLs: Extended-spectrum beta-lactamases (ESBLs) are enzymes produced by Providencia stuartii that confer resistance to beta-lactam antibiotics, including penicillins, cephalosporins, and monobactams. These ESBLs are often encoded by plasmids, allowing for the horizontal transfer of antibiotic-resistance genes among bacteria.
Integrons: Providencia stuartii can harbor integrons, which are genetic elements capable of capturing and expressing antibiotic resistance genes from various sources. Integrons play a significant role in conferring resistance to multiple classes of antibiotics, such as aminoglycosides, fluoroquinolones, sulfonamides, and trimethoprim.
Strains of Providencia stuartii:
P. stuartii MRSN 2154: This strain of Providencia stuartii was isolated from a wound infection in the United States. With a genome size of 4.7 Mb and containing 4,318 protein-coding genes, it carries multiple antibiotic resistance genes, such as blaTEM-1, blaSHV-12, blaOXA-9, blaNDM-1, and aac(6′)-Ib-cr.
P. stuartii RYC492: Isolated from human urine in Spain, this strain has a genome size of 4.6 Mb and contains 4,232 protein-coding genes.
P. stuartii ATCC 25827: Isolated from human feces in the United States, this strain has a genome size of 4.5 Mb and contains 4,122 protein-coding genes.
P. stuartii DSM 3043: Isolated from human urine in Germany, this strain has a genome size of 4.5 Mb and contains 4,137 protein-coding genes.
Providencia stuartii employs several fundamental mechanisms to initiate and establish infections in the host. Adhesion and colonization are facilitated by specific outer membrane proteins like fimbriae, pili, and lipopolysaccharides, allowing the bacterium to adhere to various host tissues, such as those in the urinary tract, respiratory tract, skin, or wounds.
Once adhered, Providencia stuartii can invade host epithelial cells and escape from phagolysosomes using enzymes like phospholipase A, hemolysin, and protease. These enzymes damage host membranes, leading to cell lysis, inflammation, and necrosis.
Motility, enabled by peritrichous flagella, allows Providencia stuartii to swarm on semi-solid surfaces and efficiently colonize host tissues. Additionally, the bacterium forms biofilms that protect host immune responses and antimicrobial agents, leading to persistent infections.
Antibiotic resistance is a significant concern with Providencia stuartii, as it can produce extended-spectrum beta-lactamases (ESBLs) that hydrolyze beta-lactam antibiotics, rendering them ineffective. Furthermore, Providencia stuartii can harbor integrons that capture and express antibiotic-resistance genes, leading to resistance against multiple classes of antibiotics.
Another tactic Providencia stuartii uses involves the modulation of porins, which affects the outer membrane’s permeability and can influence the bacterium’s susceptibility to antibiotics. In severe cases or immunocompromised individuals, Providencia stuartii can disseminate from the initial infection site to other body parts through the bloodstream, leading to systemic infections like pneumonia or meningitis.
In the battle against Providencia stuartii, the innate immune system deploys an array of robust mechanisms to halt the infection at its early stages. Macrophages and epithelial cells produce nitric oxide (NO). This potent molecule can wreak havoc on bacterial membranes and DNA, effectively hindering the growth of P. stuartii both in laboratory settings and within the host’s body.
Additionally, epithelial cells and neutrophils secrete antimicrobial peptides, such as defensins, cathelicidins, and LL-37. These act as small yet formidable proteins that disrupt bacterial membranes and interfere with their essential metabolic processes. Furthermore, the complement system, a group of serum proteins, gets activated to initiate a cascade of reactions.
This process culminates in forming membrane attack complexes (MACs) on the bacterial surface. MACs bore holes into the bacterial membrane, inducing cell lysis and destruction. Additionally, the complement system enhances the process of phagocytosis, whereby immune cells engulf and dispose of the bacteria, further fortifying the body’s defenses.
As the infection progresses, the adaptive immune system joins the fray, arming itself with specialized tactics to counter P. stuartii. B lymphocytes are deployed to produce antibodies that recognize and bind to specific antigens present on the surface of the bacteria. These antibodies serve many purposes – they neutralize the bacteria, facilitate phagocytosis, and enhance the opsonization process by interacting with Fc receptors on phagocytes.
Simultaneously, antigen-presenting cells activate T lymphocytes, divided into two primary subsets: helper T cells (Th) and cytotoxic T cells (Tc). Helper T cells orchestrate the immune response by releasing cytokines that regulate the activities of various immune cells, like B cells, neutrophils, & macrophages. This coordination is crucial in combating the infection effectively.
Providencia stuartii is a significant pathogen known to cause various infections, with urinary tract infections being one of the most common in catheterized patients, particularly in hospitals and long-term care facilities. However, this opportunistic pathogen can also lead to other severe infections, including wound infections, pneumonia, bacteremia, meningitis, and Fournier’s gangrene, particularly in critically ill patients or those with underlying medical conditions. Clinical manifestations of Providencia stuartii infections often include fever, tachycardia, hypotension, suprapubic tenderness, and flank pain.
In elderly individuals, who are more prone to having long-term indwelling urinary catheters, the risk of P. stuartii infections is high. The bacterium has also been found to cause neonatal diarrhea in dairy cows. In humans, Providencia stuartii can be isolated from various sources, including urine (most common), stool, blood, sputum, skin, and wound cultures. In cases of P. stuartii sepsis, the primary origin is often urinary. Notably, it is the most common cause of purple urine bag syndrome.
Physical examination of patients with Providencia stuartii bloodstream infection typically reveals symptoms like fever, rapid heart rate (tachycardia), and low blood pressure (hypotension). Due to its opportunistic nature and association with invasive medical procedures, healthcare providers must be vigilant in diagnosing and managing Providencia stuartii infections, especially in vulnerable populations.
Identifying Providencia stuartii from other bacteria, particularly within the Enterobacteriaceae family, involves a series of diagnostic tests that showcase its distinct characteristics:
Culture test: Providencia stuartii displays non-lactose fermenting properties, forming colorless colonies on MacConkey agar due to lactose fermentation’s lack of acid production.
Gram stain: Providencia stuartii is recognized as Gram-negative bacilli, exhibiting a pink color after counterstaining with safranin rather than retaining the purple dye.
Motility test: Providencia stuartii‘s motility is evident, as it possesses flagella that allow it to move in liquid media or semi-solid agar, leading to diffuse growth or turbidity in motility test medium or SIM agar.
Voges-Proskauer (VP) test: Providencia stuartii exhibits a negative VP test, as it does not produce acetoin or 2,3-butanediol from glucose fermentation, resulting in the absence of a red complex when Barritt’s reagent is applied.
Oxidase test: Providencia stuartii is characterized as oxidase negative, indicating the absence of the enzyme cytochrome c oxidase.
Indole test: Providencia stuartii exhibits a positive indole test, demonstrating its capability to produce indole from the amino acid tryptophan, resulting in a distinct red ring with Kovac’s reagent.
Methyl red (MR) test: Providencia stuartii yields a positive MR test, indicating its ability to produce mixed acids from glucose fermentation, which lowers the medium’s pH below 4.4, causing a color shift from yellow to red with methyl red.
Urease test: Providencia stuartii tests negative for urease production, indicating the absence of hydrolysis of urea to ammonia and carbon dioxide, thus preventing a color shift from yellow to pink on urea agar or broth.

Both our subscription plans include Free CME/CPD AMA PRA Category 1 credits.

On course completion, you will receive a full-sized presentation quality digital certificate.
A dynamic medical simulation platform designed to train healthcare professionals and students to effectively run code situations through an immersive hands-on experience in a live, interactive 3D environment.
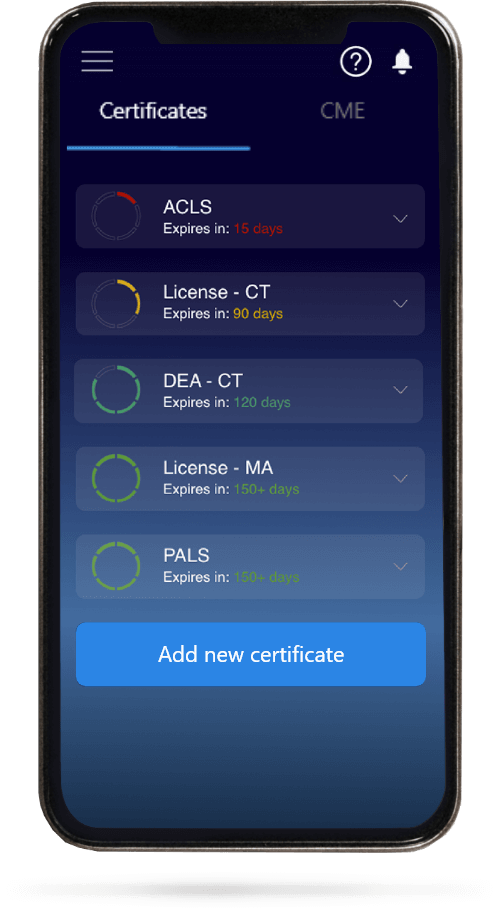
When you have your licenses, certificates and CMEs in one place, it's easier to track your career growth. You can easily share these with hospitals as well, using your medtigo app.